Introduction | Peering into the Heart of the Atom
Hello students! In our previous discussions on atoms, we learned that an atom is mostly just empty space. If you were to imagine an atom enlarged to the size of your entire classroom, the nucleus sitting at its center would be no bigger than the tiny head of a pin!
However, don’t let its microscopic size fool you. Despite being roughly $10^{4}$ times smaller in radius than the atom itself, this tiny central core is densely packed. It holds more than 99.9% of the entire atom’s mass! In this chapter, we are going to shift our focus away from the orbiting electrons and dive straight into the nucleus. We will explore what it is made of, the invisible forces that hold it together, and the massive amounts of energy it can release. Let’s get started!
1. Atomic Masses and the Composition of the Nucleus
1.1 The Atomic Mass Unit (u)
When we talk about the mass of a nucleus, using standard kilograms is incredibly inconvenient. For instance, the mass of a single carbon atom is a minuscule $1.992647\times10^{-26}$ kg. To make our lives easier, physicists defined a special unit called the atomic mass unit (u).
By definition, 1 atomic mass unit (1 u) is exactly $1/12^{th}$ of the mass of a single Carbon-12 (${}^{12}C$) atom. When we calculate this in kilograms, we get:
1.2 Protons and Neutrons
So, what lives inside this tiny nucleus? Two types of particles, collectively called nucleons.
- Protons: These carry a positive fundamental charge. The number of protons defines the element and is known as the atomic number ($Z$). The mass of a proton is $m_{p}=1.00727u$.
- Neutrons: Discovered by James Chadwick in 1932 (who won a Nobel Prize for it in 1935!), neutrons are neutral particles. They have almost the same mass as a proton, specifically $m_{n}=1.00866u$. Inside the nucleus, a neutron is stable, but outside, a free neutron decays in about 1000 seconds.
Notation: We represent a specific nuclear species (nuclide) using the notation ${}_{Z}^{A}X$. Here, $X$ is the chemical symbol, $Z$ is the atomic number (number of protons), and $A$ is the mass number (total protons + neutrons).
1.3 Isotopes, Isobars, and Isotones
When we measure atomic masses accurately using a mass spectrometer, we notice something fascinating: not all atoms of the same element weigh the same.
- Isotopes: Atoms of the same element (same $Z$) but with different mass numbers ($A$) because they have a different number of neutrons. Since chemical properties depend on electrons (and therefore $Z$), isotopes behave identically in chemical reactions. For example, Chlorine has two main isotopes: 34.98 u (75.4% abundance) and 36.98 u (24.6% abundance). The average mass is 35.47 u.
- Isobars: Nuclides from different elements that happen to have the same mass number ($A$). For example, ${}_{1}^{3}H$ (Tritium) and ${}_{2}^{3}He$ are isobars.
- Isotones: Nuclides with the same number of neutrons ($N$) but different atomic numbers ($Z$).
2. Size and Density of the Nucleus
How big is a nucleus, and how densely packed is it?
Pioneering experiments involving the scattering of fast electrons have allowed us to measure the sizes of various nuclei very accurately. We find that the nucleus behaves almost like a drop of liquid. Its volume is directly proportional to its mass number ($A$).
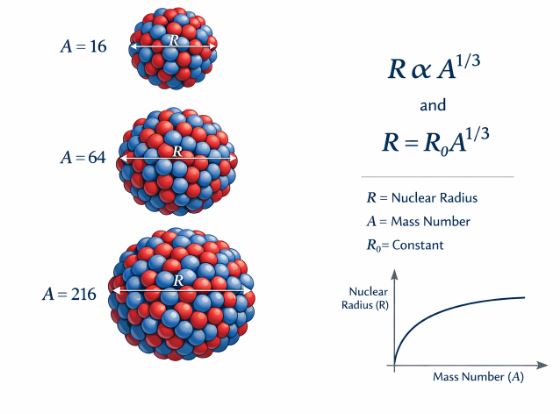
Figure-1: The radius of a nucleus increases with the cube root of its mass number.
The Radius Formula:
Where $R_{o}$ is a constant empirically found to be $1.2\times10^{-15}m$ (or 1.2 fm).
Teacher’s Insight on Density: If the volume is proportional to $A$, and the mass is roughly proportional to $A$, what happens to density? Density is Mass/Volume. This means the $A$ cancels out! The density of nuclear matter is a constant, independent of the size of the nucleus. This density is incredibly high, roughly $2.3\times10^{17}kg~m^{-3}$. To put this in perspective, this is comparable to the density of neutron stars out in space!
3. Mass-Energy Equivalence and Mass Defect
3.1 Einstein’s Famous Equation
Before Albert Einstein, scientists believed that mass and energy were two entirely separate concepts that were conserved independently. Einstein’s theory of special relativity revolutionized this by proving that mass is just another form of energy.
Here, $c$ is the speed of light ($3\times10^{8}m~s^{-1}$). Because $c^{2}$ is such a massive number, converting even 1 gram of matter completely into energy would release $9\times10^{13}J$ of energy!
3.2 The Mystery of the Missing Mass (Mass Defect)
If you take 8 protons and 8 neutrons and place them on a scale, you get a certain total mass. But if you combine them to form an Oxygen-16 (${}_{8}^{16}O$) nucleus, the mass of the nucleus is less than the sum of its individual parts!
This difference is called the Mass Defect ($\Delta M$).
Where did the missing mass go? According to Einstein, it was converted into energy to bind the nucleus together. If we ever wanted to rip that Oxygen nucleus back into individual protons and neutrons, we would have to supply exactly that much energy back. We call this the Nuclear Binding Energy ($E_{b}$).
Pro Tip for Exams: The energy equivalent of 1 atomic mass unit (1 u) is roughly 931.5 MeV.
4. Binding Energy per Nucleon
Total binding energy isn’t the best way to judge how stable a nucleus is. A heavier nucleus will naturally have a larger total binding energy simply because it has more parts. A better metric is the Binding Energy per Nucleon ($E_{bn}$), which is the average energy required to remove a single nucleon from the nucleus.
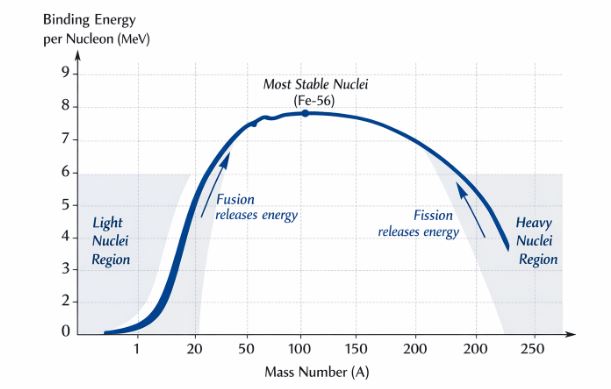
Figure-2: The Binding Energy curve. Notice the plateau in the middle and the drop-offs at both the light and heavy ends.
Key Observations from the Curve (Crucial for Boards):
- For intermediate nuclei ($30 < A < 170$), the curve is relatively flat and constant at about 8.0 MeV per nucleon. The maximum stability peaks near Iron ($A=56$) at about 8.75 MeV.
- For light nuclei ($A < 30$) and heavy nuclei ($A > 170$), the binding energy per nucleon drops noticeably.
What does this mean? Nature favors stability (higher $E_{bn}$).
- If a very heavy nucleus splits into two lighter ones, the new fragments jump up into the more stable plateau region. This releases energy and is called Nuclear Fission.
- If two very light nuclei combine, the new heavier nucleus also climbs up toward the stable plateau. This also releases energy and is called Nuclear Fusion.
5. The Strong Nuclear Force
Let’s address the elephant in the room: Protons are positively charged. Like charges repel. So why doesn’t a nucleus, packed with protons, instantly explode due to Coulomb repulsion?
There must be another force at play—one that is overwhelmingly stronger than the Coulomb electrostatic force. This is the Strong Nuclear Force. Here are its unique properties:
- Incredibly Strong: It easily dominates the repulsive Coulomb force between protons.
- Extremely Short-Ranged: It only operates over a distance of a few femtometers. Beyond that, it falls rapidly to zero.
- Saturation Property: Because it is short-ranged, a nucleon only interacts with its nearest neighbors. This is why the binding energy per nucleon remains constant for intermediate-mass nuclei.
- Charge Independent: The force between neutron-neutron, proton-proton, and neutron-proton is essentially identical.
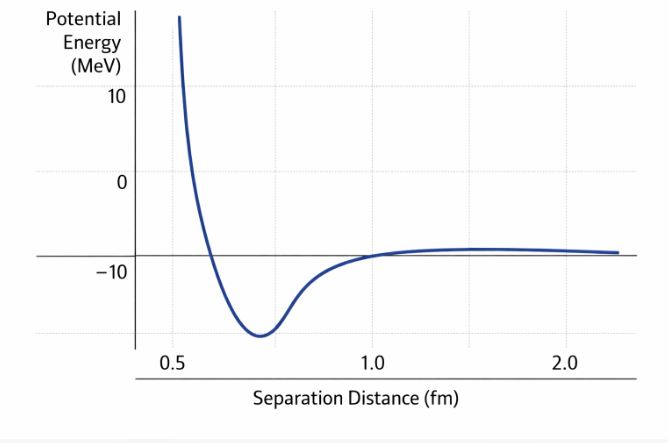
Figure-3: Potential energy between two nucleons. It reaches a minimum at about 0.8 fm. Below this, the force becomes strongly repulsive, preventing the nucleus from collapsing in on itself.
6. Radioactivity (A Quick Overview)
Discovered purely by accident in 1896 by A.H. Becquerel while studying uranium-potassium sulphate and photographic plates, radioactivity is a nuclear phenomenon. An unstable nucleus will spontaneously undergo decay to achieve a more stable state. Nature provides three primary types of radioactive decay:
- $\alpha$-decay: Emission of a Helium nucleus (${}_{2}^{4}He$).
- $\beta$-decay: Emission of electrons or positrons.
- $\gamma$-decay: Emission of high-energy electromagnetic photons.
7. Harnessing Nuclear Energy
In traditional chemical reactions (like burning coal), the energy released comes from electron rearrangements and is measured in mere electron volts (eV). However, in nuclear reactions, the energy released is measured in millions of electron volts (MeV). Fission of 1 kg of uranium generates $10^{14}$ J of energy, compared to just $10^{7}$ J from 1 kg of coal!
7.1 Nuclear Fission
Fission occurs when a heavy nucleus is bombarded by a neutron and splits into intermediate-mass fragments. A classic example:
This reaction releases an enormous amount of energy—roughly 200 MeV per uranium nucleus. The energy first appears as the kinetic energy of the fragments and neutrons, eventually transferring to surrounding matter as heat. This heat is harnessed in nuclear power reactors to produce electricity.
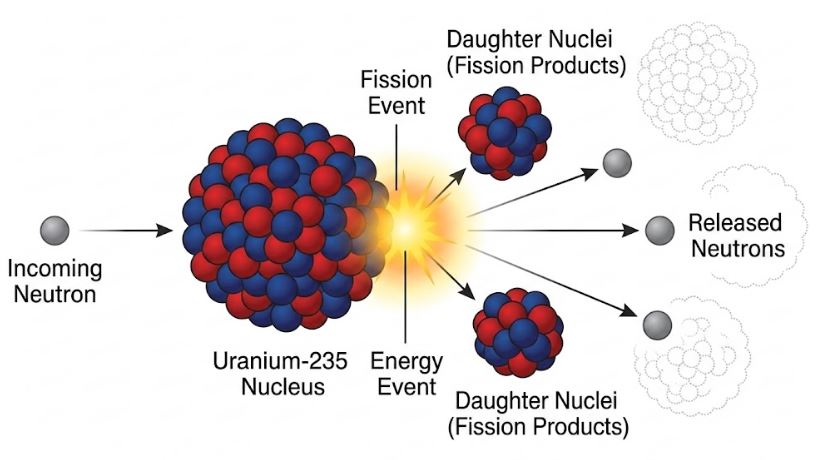
Figure-4: Fission of Uranium-235. Notice how one incoming neutron results in multiple outgoing neutrons, which can trigger a chain reaction.
7.2 Nuclear Fusion (The Power of the Stars)
Fusion is the opposite of fission. Two light nuclei combine to form a heavier, more stable nucleus, releasing energy. However, getting two positively charged nuclei close enough for the short-range strong nuclear force to grab them is tough due to Coulomb repulsion. They must have massive kinetic energy, which requires extreme temperatures (like $10^{9} K$). This is why it’s called thermonuclear fusion.
This process powers the Sun and all stars. In our Sun, hydrogen is constantly being “burned” into helium through a multi-step process called the proton-proton (p-p) cycle. The net result is that 4 hydrogen atoms combine to form a helium atom, releasing 26.7 MeV of energy.
Real-Life Examples (CBSE Context)
- Medical Diagnostics and Treatment: Unstable isotopes created artificially are used extensively in medicine. For instance, radioactive iodine is used to image and treat thyroid conditions.
- Carbon Dating: Archeologists use the known half-life of Carbon-14 (an unstable isotope) to determine the age of ancient artifacts and fossils.
- Nuclear Power Plants: India operates several nuclear reactors (like at Tarapur and Kudankulam) that use controlled nuclear fission to boil water, spin turbines, and generate electricity.
- The Sun’s Longevity: The Sun has been fusing hydrogen for 5 billion years and has enough fuel to continue for another 5 billion years before it cools and expands into a red giant.
- Future Tech – Fusion Reactors: Scientists in India and worldwide are trying to build controlled fusion reactors. Because the plasma must reach $10^{8} K$, no physical container can hold it. They use strong magnetic fields to confine the plasma. If successful, this will provide almost unlimited clean energy.
Common Student Misconceptions
1. “Mass is conserved in nuclear reactions.”
Correction: Strictly speaking, mass alone is not conserved. Rest mass is converted into energy (or vice-versa). We must treat mass and energy as a single unified entity: Mass-Energy. What *is* strictly conserved is the number of nucleons (protons and neutrons).
2. “$E=mc^{2}$ only applies to nuclear reactions.”
Correction: Mass-energy interconversion technically happens in standard chemical reactions too! The heat released in a chemical reaction comes from chemical mass defect. However, because chemical energies are millions of times smaller than nuclear energies, the mass change in chemistry is far too tiny to measure.
CBSE-Aligned Practice Set with Solutions
Very Short Answer Questions (1 Mark)
Q1. Why is the density of a nucleus independent of its mass number?
Solution: The radius of a nucleus is $R = R_{o}A^{1/3}$. Therefore, the volume ($V \propto R^{3}$) is proportional to the mass number $A$. Since both mass and volume are proportional to $A$, the ratio (density) remains constant.
Q2. Name the elementary particle emitted along with a proton and electron during the decay of a free neutron.
Solution: An antineutrino.
Q3. Two nuclei have mass numbers in the ratio 1:8. What is the ratio of their nuclear radii?
Solution: $R_{1}/R_{2} = (A_{1}/A_{2})^{1/3} = (1/8)^{1/3} = 1/2$. The ratio is 1:2.
Short Answer Questions (2-3 Marks)
Q4. Explain why the binding energy per nucleon curve drops for heavy nuclei ($A > 170$). What phenomenon does this lead to?
Solution: For heavy nuclei, the large number of protons creates a massive Coulomb repulsive force. Because the strong nuclear force is short-ranged and saturates, it cannot compensate for the long-range electrostatic repulsion across the large diameter of the nucleus. This makes the nucleus less tightly bound, causing the curve to drop. This instability leads to radioactive decay and makes them susceptible to nuclear fission.
Q5. Distinguish between isotopes and isobars with one example of each.
Solution: Isotopes are atoms of the same element with the same atomic number ($Z$) but different mass numbers ($A$). Example: ${}_{1}^{2}H$ (Deuterium) and ${}_{1}^{3}H$ (Tritium). Isobars are atoms of different elements having the same mass number ($A$) but different atomic numbers ($Z$). Example: ${}_{1}^{3}H$ and ${}_{2}^{3}He$.
Q6. Calculate the energy equivalent of 1 atomic mass unit (1 u) in MeV. (Given 1 u = $1.66 \times 10^{-27}$ kg, c = $3 \times 10^{8}$ m/s)
Solution:
$E = mc^{2} = (1.66 \times 10^{-27} kg) \times (3 \times 10^{8} m/s)^{2} \approx 1.49 \times 10^{-10} J$.
To convert Joules to eV, divide by $1.6 \times 10^{-19}$ C.
$E = (1.49 \times 10^{-10}) / (1.6 \times 10^{-19}) = 931.5 \times 10^{6} eV = 931.5 MeV$.
Long Answer Questions (5 Marks)
Q7. (a) Define nuclear binding energy and mass defect. (b) Draw the plot of binding energy per nucleon versus mass number. (c) Use this graph to explain the release of energy in both nuclear fission and nuclear fusion.
Solution:
(a) Mass Defect ($\Delta M$): It is the difference between the sum of the masses of individual nucleons (protons and neutrons) and the actual mass of the nucleus. $\Delta M = [Zm_{p} + (A-Z)m_{n}] – M$.
Binding Energy: The energy equivalent of the mass defect. It is the energy required to break a nucleus completely into its constituent nucleons. $E_{b} = \Delta M c^{2}$.
(b) [Students must draw Figure-2 from notes, showing the peak at Fe-56, flat region, and drops at both ends].
(c) Fission: A heavy nucleus ($A \approx 240$) has a lower binding energy per nucleon (~7.6 MeV). When it splits into two intermediate nuclei ($A \approx 120$), the nucleons become more tightly bound (~8.5 MeV). This transition releases the difference in energy.
Fusion: Very light nuclei ($A < 10$) have very low binding energy per nucleon. When they combine to form a heavier nucleus, the system moves up the curve to a state of higher binding energy, resulting in a net release of energy.
Q8. State the key properties of the strong nuclear force. How does the concept of ‘saturation’ of nuclear forces explain the constancy of binding energy per nucleon in the range $30 < A < 170$?
Solution:
Properties: (i) Strongest interaction in nature. (ii) Extremely short-ranged (few fm). (iii) Charge independent (p-p, n-n, p-n forces are roughly equal). (iv) Becomes strongly repulsive below 0.8 fm.
Saturation Explanation: Because the nuclear force is short-ranged, a nucleon deep inside a large nucleus only interacts with its immediate neighboring nucleons, up to a maximum number $p$. Adding more nucleons to the atom increases its size, but does not add to the binding energy of that internal nucleon because the new nucleons are too far away to exert a nuclear force on it. Since most nucleons in intermediate/large nuclei sit in the interior, the average binding energy per nucleon remains constant, proportional to this limited number of neighbors. This property is called saturation.
Case-Based / Competency-Based Question (4 Marks)
Q9. The source of energy of stars is nuclear fusion. The interior of the sun has a core temperature of about $1.5 \times 10^{7} K$. Hydrogen is the fuel in the sun’s core, which is burned into helium via the proton-proton (p-p) cycle. It is estimated that the sun has been fusing hydrogen for 5 billion years and has enough fuel for another 5 billion years.
(a) Why is a very high temperature required for nuclear fusion? (1)
(b) What is the net nuclear reaction equation for the hydrogen fusion in the Sun? (1)
(c) What will happen to the Sun once its hydrogen core is depleted? (2)
Solution:
(a) High temperature provides the nuclei with sufficient kinetic energy to overcome the massive Coulomb electrostatic repulsion between the positively charged protons.
(b) Net reaction: $4{}_{1}^{1}H + 2e^{-} \rightarrow {}_{2}^{4}He + 2v + 6\gamma + 26.7 MeV$.
(c) Once hydrogen is depleted and becomes helium, the core cools and starts to collapse under its own gravity. This collapse drastically raises the core temperature again (to about $10^{8} K$), triggering the fusion of helium into carbon. The outer envelope of the sun will expand, turning it into a red giant.
Assertion-Reason Question (1 Mark)
Q10. Directions: Choose the correct option.
Assertion (A): The mass of an isolated atom of ${}_{8}^{16}O$ is exactly equal to the sum of the masses of 8 protons, 8 neutrons, and 8 electrons.
Reason (R): Mass defect in an oxygen nucleus is converted into nuclear binding energy.
A) Both A and R are true, and R is the correct explanation of A.
B) Both A and R are true, but R is NOT the correct explanation of A.
C) A is true, but R is false.
D) A is false, but R is true.
Solution: (D). The assertion is false because the mass of an oxygen atom is *less* than the sum of its constituent parts. This difference is the mass defect, which is converted to binding energy (Reason is true).
End of Notes.
Students, make sure you memorize the $E_{bn}$ vs $A$ curve and the properties of the strong nuclear force, as these are heavily tested in the board exams. Keep studying hard!
Read Also:
Class-12 Chapter 12- Atoms
For official syllabus and textbooks, visit the
NCERT Official Website.

