Introduction | Journey into the Microscopic World
Hello students! Welcome back to our Physics class. Up until the nineteenth century, scientists had gathered a substantial amount of evidence pointing towards the atomic hypothesis of matter. But what exactly was inside these atoms?
The major breakthrough came in 1897 when the brilliant English physicist J. J. Thomson conducted experiments on electric discharge through gases. His work proved a startling fact: atoms of all different elements contain identical, negatively charged constituents, which we now call electrons. However, since we know that atoms are electrically neutral as a whole, common sense tells us there must be some positive charge hidden inside to balance out these negative electrons.
This sparked the ultimate scientific mystery: How are the positive charges and electrons arranged inside the atom? What does the internal architecture of an atom actually look like? Let’s explore how humanity unraveled the structure of the atom.
1. The First Attempt: Thomson’s Plum Pudding Model
In 1898, J. J. Thomson proposed the very first model of the atom. Imagine a spherical cloud where the positive charge is uniformly distributed throughout its entire volume. Inside this positive sphere, negatively charged electrons are embedded.
Teacher’s Analogy: Think of a watermelon! The red, fleshy part represents the uniform positive charge, and the black seeds stuck inside are the electrons. Historically, this was picturesquely called the “plum pudding model”.
While elegant, this model was short-lived. Subsequent scientific studies and experiments proved that the actual distribution of positive and negative charges in an atom is drastically different from a uniform sphere. Furthermore, Thomson’s model was electrostatically unstable.
2. The Turning Point: Alpha-Particle Scattering Experiment
This is a highly important topic for your board exams. Pay close attention to the experimental setup and the shocking observations!
2.1 The Setup
In 1906, Ernst Rutherford proposed a classic experiment: firing alpha ($\alpha$) particles at atoms to investigate their internal structure. At his suggestion, in 1911, H. Geiger and a 20-year-old undergraduate named E. Marsden performed the famous scattering experiment.
- The Source: They used a radioactive Bismuth ($^{214}\text{Bi}$) source that emitted a beam of 5.5 MeV $\alpha$-particles. (Remember, an $\alpha$-particle is a helium nucleus, meaning it carries a $+2e$ charge and has the mass of a helium atom).
- Collimation: The particles passed through lead bricks to form a narrow, focused beam.
- The Target: This beam was directed at an incredibly thin gold foil, just $2.1 \times 10^{-7} \text{ m}$ thick.
- The Detector: To see where the particles went, they used a rotatable detector with a zinc sulphide (ZnS) screen and a microscope. When an $\alpha$-particle struck the screen, it produced a tiny brief flash of light (a scintillation). By rotating the microscope, they could count the number of particles scattered at various angles.
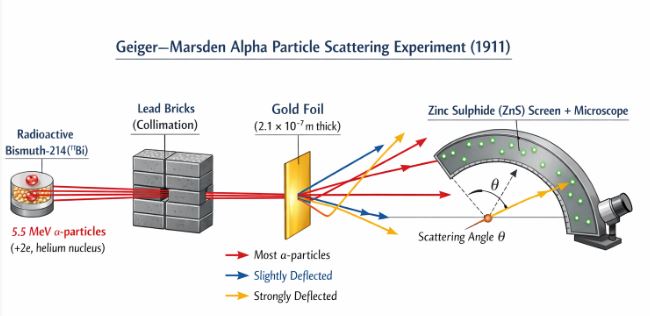
Figure-1: Schematic arrangement of the Geiger-Marsden $\alpha$-particle scattering experiment. Notice how the detector can rotate to capture particles deflected at various angles $\theta$.
2.2 The Shocking Observations
If Thomson’s model was correct, the $\alpha$-particles should have easily pierced through the uniform “pudding” with almost no deflection. But the data showed something entirely different:
- Most pass straight through: A vast majority of the $\alpha$-particles suffered no collisions and passed right through the gold foil.
- Small deflections: Only about $0.14\%$ of the incident particles were scattered by more than $1^{\circ}$.
- Large deflections (The shocker!): About 1 in 8000 $\alpha$-particles were deflected by an angle greater than $90^{\circ}$, essentially bouncing backward!.
2.3 Rutherford’s Conclusion
Rutherford argued that to make a fast, massive $\alpha$-particle completely reverse its direction, it must have experienced an incredibly strong repulsive force. This intense repulsion could only happen if the majority of the atom’s mass and its entire positive charge were tightly concentrated in a tiny central region.
Thus, the concept of the nucleus was born!. An incoming $\alpha$-particle heading straight for this dense nucleus would get very close, experience massive repulsion without penetrating it, and rebound backwards ($\theta \cong \pi$).
3. Rutherford’s Nuclear Model of the Atom
Based on the scattering data, Rutherford proposed his planetary model (nuclear model) of the atom.
- The entire positive charge and almost all the mass of the atom reside in a very small central volume called the nucleus.
- The electrons revolve around this nucleus in orbits, much like planets revolve around the sun.
- The size of the nucleus is estimated to be incredibly tiny: about $10^{-15} \text{ m}$ to $10^{-14} \text{ m}$.
- Since the overall size of an atom is about $10^{-10} \text{ m}$, the atom is roughly $10,000$ to $100,000$ times larger than its nucleus!.
Teacher’s Insight: To put this in perspective, if the nucleus were the size of a tiny seed in the center of a football field, the electrons would be buzzing around the outer edges of the stadium! Most of an atom is simply empty space. This perfectly explains why most $\alpha$-particles passed straight through the metal foil without hitting anything.
3.1 The Fatal Flaw of Rutherford’s Model
As brilliant as the nuclear model was, it had severe theoretical problems. In the classical picture, the electron orbits the nucleus. To stay in a circular orbit, the electron must be constantly accelerated toward the center (centripetal acceleration).
However, according to classical electromagnetic theory, any accelerating charged particle must emit energy in the form of electromagnetic radiation. Therefore, a revolving electron should continuously lose energy. As it loses energy, its orbit would shrink, causing it to spiral inward and eventually crash into the nucleus.
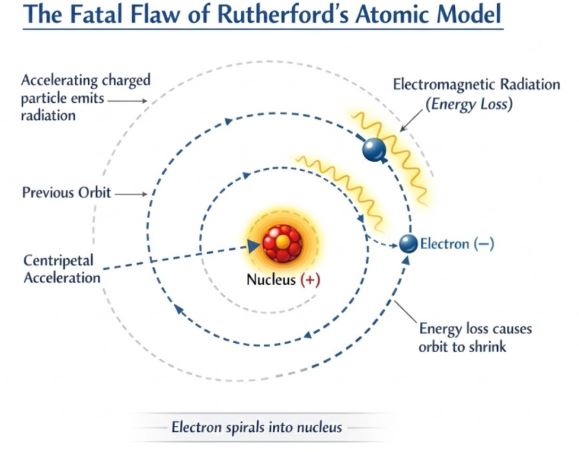
Figure-2: The classical dilemma: An accelerated orbiting electron should continuously radiate energy and spiral into the nucleus, contradicting the known stability of matter.
Calculations showed this collapse would happen in a fraction of a second. But we know atoms are stable! Matter doesn’t just collapse out of existence. Furthermore, as the electron spirals inward, its angular velocity changes continuously, which means the frequency of emitted light would change continuously, producing a continuous spectrum. But experiments showed that gases emit a discrete line spectrum. Rutherford’s model could not explain this.
4. Atomic Spectra: The Fingerprints of Elements
When we heat solids, liquids, or dense gases, they emit continuous electromagnetic radiation (a continuous spectrum containing many wavelengths) because the atoms interact heavily with their neighbors.
However, if we take a rarefied gas (like in a neon sign) and excite it electrically, it emits light of only certain specific, discrete wavelengths. The spectrum looks like a series of bright lines against a dark background, known as an emission line spectrum. Because the atoms in a gas are far apart, this radiation represents the internal behavior of individual atoms, free from neighboring interactions.
Every element has its own unique set of spectral lines. Studying these emission line spectra serves as a definitive “fingerprint” to identify the gas. Conversely, if white light passes through a cool gas, we see an absorption spectrum—dark lines on a continuous background, exactly at the frequencies the gas would normally emit.
5. Bohr’s Quantized Model of the Hydrogen Atom
To fix the stability issue and explain atomic spectra, Niels Bohr stepped in. He realized that classical physics rules simply do not apply at the microscopic atomic scale. In 1913, he boldly combined classical mechanics with the newly emerging quantum theory proposed by Planck and Einstein.
Bohr formulated three revolutionary postulates for hydrogenic (single-electron) atoms:
Postulate 1: Stationary Orbits
An electron in an atom can revolve in certain specific, dynamically stable circular orbits without emitting any radiant energy. This directly defied classical electromagnetic theory. In these allowed states, the atom is stable, and each state has a definite total energy (called stationary states).
Postulate 2: Quantization of Angular Momentum
The electron cannot just orbit at any random distance. It revolves only in those specific orbits where its angular momentum ($L$) is an integral multiple of $\frac{h}{2\pi}$.
$$L = \frac{nh}{2\pi}$$
where $h$ is Planck’s constant ($6.6 \times 10^{-34} \text{ J s}$) and $n = 1, 2, 3…$ is the principal quantum number. This mathematically locks the electron into discrete orbital tracks.
Postulate 3: Energy Transitions
While orbiting in a stationary state, an electron does not radiate energy. However, it can make a transition (jump) from a specified higher energy non-radiating orbit ($E_i$) to a lower energy orbit ($E_f$). When it drops down, it sheds the excess energy by emitting a single photon.
The frequency ($\nu$) of this emitted photon is precisely governed by the energy difference:
$$h\nu = E_i – E_f$$
. Because the allowed energy states ($E_i$, $E_f$) are discrete integers, the emitted photons can only have specific discrete frequencies, perfectly explaining why we see separated lines in an emission spectrum!.
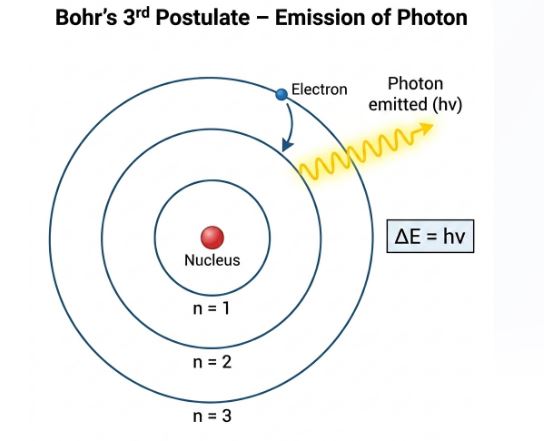
Figure-3: Bohr’s 3rd Postulate: An electron emits a photon of specific frequency when transitioning from a higher energy level to a lower one.
6. Energy Levels and Orbital Radii
By applying Coulomb’s law for electrostatic attraction ($F_e$) acting as the centripetal force ($F_c$), and substituting Bohr’s quantization condition, we can find the radius of the $n^{th}$ orbit in a hydrogen atom:
$$r_n = \left(\frac{n^2}{m}\right)\left(\frac{h}{2\pi}\right)^2 \frac{4\pi\epsilon_0}{e^2}$$
.
More importantly, the total energy ($E_n$) of the electron in the $n^{th}$ orbit is completely quantized. The formula is:
$$E_n = -\frac{13.6}{n^2} \text{ eV}$$
.
Understanding the Energy States
- Negative Sign: The total energy of the electron is negative, which signifies that the electron is physically bound to the positive nucleus. It cannot escape on its own.
- Ground State ($n=1$): The lowest state of the atom where the electron revolves in the smallest orbit. For hydrogen, the ground state energy is $-\frac{13.6}{1^2} = -13.6 \text{ eV}$. At room temperature, most hydrogen atoms naturally sit in this state.
- Excited States ($n > 1$): If the atom absorbs energy (via collision or absorbing a photon), the electron jumps to a higher orbit ($n=2, 3…$). These are excited states. The energy required to reach the first excited state ($n=2$) is $E_2 – E_1 = -3.40 \text{ eV} – (-13.6 \text{ eV}) = 10.2 \text{ eV}$.
- Ionisation Energy: The minimum energy needed to completely tear the electron away from the ground state ($n=1$) to infinity ($n=\infty$). Since $E_\infty = 0 \text{ eV}$, you must supply $+13.6 \text{ eV}$ to free the electron.

Figure-4: Energy level diagram for the hydrogen atom. Notice how the energy levels get packed closer together as the principal quantum number $n$ increases.
7. De Broglie’s Explanation of Bohr’s Second Postulate
While Bohr’s model was a brilliant success, it left one major conceptual puzzle: Why should the angular momentum be quantized as integral multiples of $\frac{h}{2\pi}$?. Bohr just assumed it to make the math work.
In 1923, French physicist Louis de Broglie solved this mystery by bringing in the wave-particle duality. He argued that the electron orbiting the nucleus must be viewed as a particle wave.
The Concept: Just like plucking a guitar string creates standing waves where only specific wavelengths survive (those with nodes at the ends), an electron wave can only persist in a circular orbit if it forms a resonant standing wave. This means the total circumference of the orbit must perfectly fit a whole integer number of wavelengths. If it doesn’t, the wave interferes with itself and its amplitude drops to zero.
Mathematically, the circumference equals $n$ times the wavelength ($\lambda$):
$$2\pi r_n = n\lambda \quad (n = 1, 2, 3…)$$
.
From de Broglie’s formula, we know wavelength is related to momentum ($p = mv_n$) as $\lambda = \frac{h}{mv_n}$. Substituting this in:
$$2\pi r_n = \frac{nh}{mv_n}$$
Rearranging the terms, we get:
$$mv_n r_n = \frac{nh}{2\pi}$$
. Since $mv_n r_n$ is angular momentum ($L$), this perfectly derives Bohr’s quantum condition!.
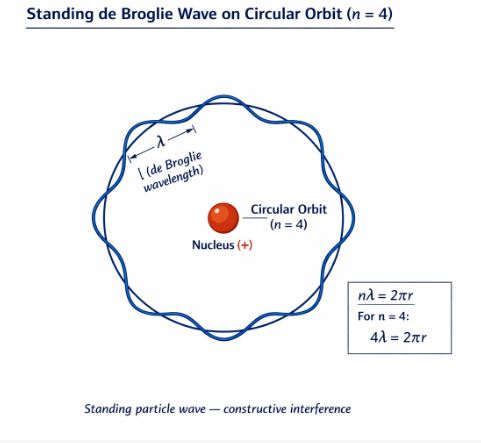
Figure-5: A standing particle wave on a circular orbit where $n=4$. The circumference perfectly holds four de Broglie wavelengths.
8. Limitations of Bohr’s Model
Science never stops evolving. While Bohr’s model gave us a fantastic framework, it had noticeable limitations that required the invention of modern Quantum Mechanics to fix:
- Limited to Hydrogenic Atoms: Bohr’s model only successfully analyzes atoms with a single electron (like Hydrogen, $He^+$, $Li^{++}$). When scientists tried to apply it to a mere two-electron atom like neutral Helium, the model completely failed. This is because Bohr’s math only considers the electrical force between the electron and the nucleus. It completely ignores the complex electron-electron electrical repulsion that occurs in multi-electron atoms.
- Cannot Explain Spectral Line Intensities: While the model correctly predicts the specific frequencies (colors) of light emitted, it cannot explain why some emission lines are intensely bright while others are very faint. Experimental observations show that certain atomic transitions are “favored” over others, a phenomenon Bohr’s model simply cannot account for.
9. Solved Numericals (Teacher’s Walkthrough)
Numerical 1 (Distance of Closest Approach):
Question: In a Geiger-Marsden experiment, an $\alpha$-particle with kinetic energy 7.7 MeV approaches a gold nucleus ($Z=79$). What is the distance of closest approach before it momentarily rests and reverses direction?
Solution:
The key idea here is conservation of mechanical energy. As the $\alpha$-particle approaches the nucleus, its kinetic energy ($K$) converts entirely into electrostatic potential energy ($U$) at the stopping point (distance $d$).
Initial Energy $E_i = K = 7.7 \text{ MeV} = 7.7 \times 1.6 \times 10^{-13} \text{ J} = 1.2 \times 10^{-12} \text{ J}$.
Final Energy $E_f = U = \frac{1}{4\pi\epsilon_0}\frac{(2e)(Ze)}{d}$.
Equating $K = U$, we get $d = \frac{2Ze^2}{4\pi\epsilon_0 K}$.
Substituting values: $d = \frac{9 \times 10^9 \times 2 \times 79 \times (1.6 \times 10^{-19})^2}{1.2 \times 10^{-12}}$.
$d = 3.0 \times 10^{-14} \text{ m}$ or $30 \text{ fm}$.
Note: The $\alpha$-particle reverses motion without physically touching the gold nucleus, as the actual nuclear radius is about 6 fm.
Numerical 2 (Energy Levels and Radius):
Question: It takes 13.6 eV of energy to separate a hydrogen atom into a proton and an electron. Compute the orbital radius in this ground state.
Solution:
Given, the total energy of the electron is $E = -13.6 \text{ eV}$.
Convert to Joules: $E = -13.6 \times 1.6 \times 10^{-19} \text{ J} = -2.2 \times 10^{-18} \text{ J}$.
From the energy relation, $E = -\frac{e^2}{8\pi\epsilon_0 r}$.
Rearranging for radius: $r = -\frac{e^2}{8\pi\epsilon_0 E}$.
$r = -\frac{(9 \times 10^9) \times (1.6 \times 10^{-19})^2}{2 \times (-2.2 \times 10^{-18})}$.
$r = 5.3 \times 10^{-11} \text{ m}$. This is known as the Bohr radius!.
Real-Life Examples (CBSE Context)
- Neon Signs: The vibrant glowing advertisements you see at night are practical applications of atomic emission spectra. Electric current passing through rarefied gases excites the electrons to higher states. When they drop back down, they emit discrete visible wavelengths characteristic of the gas.
- Astrophysics & Spectroscopy: Astronomers determine the chemical composition of distant stars by analyzing the absorption spectrum of the starlight. By looking at the dark spectral lines, they identify the “fingerprints” of gases present in the star’s atmosphere.
Important Board Exam Derivations Summary
Students, please practice these derivations repeatedly as they carry heavy weightage:
- Distance of Closest Approach: Derived using the principle of conservation of mechanical energy ($K = U$).
- Radius of $n^{th}$ Bohr Orbit: Equate Coulomb force to Centripetal force ($F_e = F_c$), and substitute velocity $v$ using Bohr’s quantization condition ($L = \frac{nh}{2\pi}$).
- De Broglie’s Proof of Quantization: Equating circumference to integral wavelengths ($2\pi r = n\lambda$) and substituting $\lambda = \frac{h}{p}$ to prove $L = \frac{nh}{2\pi}$.
Multiple Choice & Practice Questions (CBSE Pattern)
- Q1. [VSA] What is the significance of the negative energy of an electron in an orbit?
Answer: The negative sign implies that the electron is bound to the positive nucleus by electrostatic attraction. To free it, external positive energy must be supplied. - Q2. [VSA] According to Rutherford’s model, what is the relative scale of the nucleus compared to the entire atom?
Answer: The atom is roughly $10,000$ to $100,000$ times larger than the nucleus. Most of the atom is empty space. - Q3. [SA] Why is Rutherford’s model unable to explain the stability of matter?
Answer: In Rutherford’s model, the electron revolves around the nucleus and possesses centripetal acceleration. According to classical electromagnetic theory, an accelerating charged particle must radiate electromagnetic waves and lose energy. Consequently, the electron would spiral inward and collapse into the nucleus, making the atom inherently unstable. - Q4. [SA] State Bohr’s quantization condition of angular momentum.
Answer: Bohr’s second postulate states that an electron can revolve only in those stable orbits where its angular momentum ($L$) is an integral multiple of $\frac{h}{2\pi}$. Mathematically, $L = \frac{nh}{2\pi}$, where $n$ is an integer ($1,2,3…$) and $h$ is Planck’s constant. - Q5. [Assertion-Reason]
Assertion (A): Bohr’s atomic model cannot be used to explain the spectrum of a neutral Helium atom.
Reason (R): Bohr’s model is only applicable to hydrogenic (single-electron) atoms because it does not account for the electrical repulsive forces between multiple electrons.
Answer: Both A and R are true, and R is the correct explanation of A. - Q6. [Case-Based] In an experiment, an electron in a hydrogen atom makes a transition from the $n=3$ excited state to the $n=1$ ground state.
(a) Is a photon absorbed or emitted? Why?
(b) Calculate the energy of the photon.
Answer:
(a) A photon is emitted because the electron is jumping from a higher energy state to a lower energy state.
(b) Energy in state $n$: $E_n = -\frac{13.6}{n^2} \text{ eV}$.
$E_1 = -13.6 \text{ eV}$.
$E_3 = -\frac{13.6}{9} = -1.51 \text{ eV}$.
Energy of emitted photon $= E_i – E_f = E_3 – E_1 = -1.51 – (-13.6) = 12.09 \text{ eV}$.
End of Notes.
Students, mastering the concepts of closest approach and energy level transitions is crucial. Keep practicing the numericals and derivations to secure top marks!
Read Also:
Class-12 Chapter 11- Dual Nature of Radiation and Matter
For official syllabus and textbooks, visit the
NCERT Official Website.

