
Simplified Notes: Class 9 Science Chapter 1 Matter in Our Surroundings Notes
Science Notes | Chapter 1 | In-Depth Analysis
Class 9 Science Chapter 1 Matter in Our Surroundings notes are explained here in simple and easy language for CBSE students.
1. What is Matter?
Matter is anything that has mass and occupies space (volume). Think about it—the book you’re reading, the water you drink, and the air you breathe are all examples of matter. Even things you can’t see, like air, are matter because they take up space and have mass.
2. The Particulate Nature of Matter
For a long time, people debated whether matter was continuous, like a solid block, or made up of tiny particles. We now know that all matter is composed of extremely small particles.
2.1 Evidence
- Evidence: When you dissolve salt in water, the salt particles spread out and fill the empty spaces between the water particles. This is why the water level doesn’t rise!
3. Characteristics of Particles of Matter
The particles that make up matter have some interesting properties:
- They have space between them: Particles are not packed together tightly; there are always gaps between them. This is why substances can be mixed or dissolved into one another.
- They are constantly moving: Particles are never still; they are always in random motion. This movement gives them kinetic energy. As you increase the temperature, the particles move faster because their kinetic energy increases.
- They attract each other: There are forces of attraction between particles that hold them together. The strength of this attraction varies from one type of matter to another. For example, it’s very strong in a solid like an iron nail but weaker in a liquid like water.
4. The Three States of Matter
Matter typically exists in three states: solid, liquid, and gas. These states exist because of the differences in how their particles are arranged and move.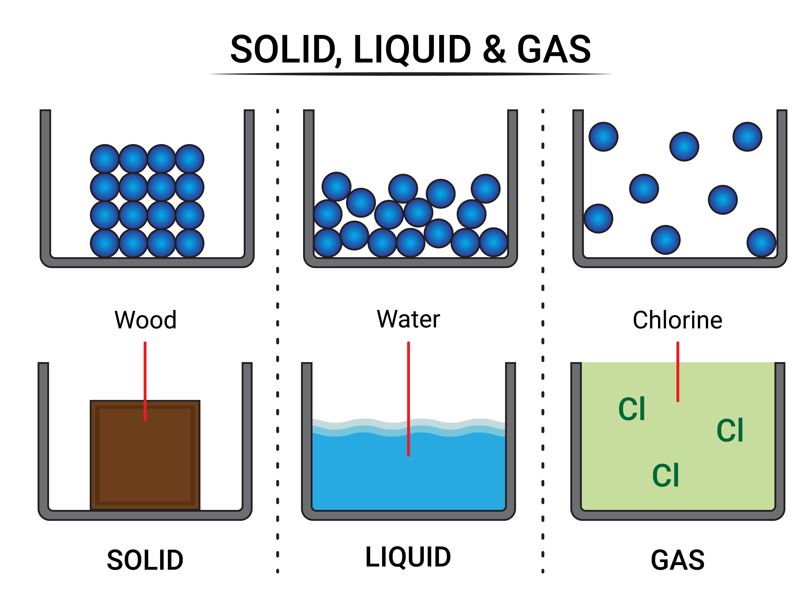
The arrangement and movement of particles determine the state of matter.
4.1 🧊 Solids
- Definite shape and volume: Solids have a fixed shape and take up a specific amount of space.
- Rigid: They are hard to bend or change shape.
- Incompressible: You can’t squeeze them into a smaller volume.
- Particle Arrangement: Particles are tightly packed with very strong forces of attraction and minimal space between them. They only vibrate in their fixed positions.
Examples: A book, a pen, or a wooden table.
4.2 💧 Liquids
- Definite volume, no definite shape: Liquids have a fixed volume but take the shape of their container.
- Fluid: They can flow easily from one container to another.
- Slightly compressible: They can be compressed a little bit.
- Particle Arrangement: Particles are loosely packed with weaker forces of attraction than solids. They can slide past one another.
Examples: Water, milk, or juice.
4.3 💨 Gases
- No definite shape or volume: Gases take both the shape and volume of their container.
- Fluid: They can flow very easily.
- Highly compressible: You can squeeze a large amount of gas into a small container. This is how LPG and CNG cylinders work.
- Particle Arrangement: Particles are far apart with very weak forces of attraction. They move around randomly and at high speeds.
Examples: Air, oxygen, or the smell of food.
5. Can Matter Change its State?
Yes! Matter can change from one state to another when you change the temperature or pressure.
Heating a substance increases the kinetic energy of its particles, causing it to change state from solid to liquid to gas.
5.1 🔥 Effect of Temperature
- Melting (Fusion): When you heat a solid (like ice), its particles gain energy and start vibrating more. Eventually, they break free from their fixed positions, and the solid turns into a liquid. The temperature at which this happens is called the melting point.
- Boiling (Vaporization): If you keep heating the liquid, the particles gain even more energy and start moving faster. At the boiling point, they have enough energy to escape into the air as a gas (or vapor).
- Condensation: This is the reverse of boiling. When a gas cools down, its particles lose energy, move slower, and turn back into a liquid. Think of water droplets forming on a cold glass.
- Freezing (Solidification): When a liquid cools, its particles slow down until they lock into fixed positions, turning it into a solid.
5.2 ✨ What is Latent Heat?
When a substance changes its state (like ice melting into water), the temperature stays the same until the change is complete, even though you are still adding heat. This “hidden” heat is called latent heat. It’s the energy used to break the bonds between particles.
- Latent Heat of Fusion: The energy needed to change a solid to a liquid at its melting point.
- Latent Heat of Vaporization: The energy needed to change a liquid to a gas at its boiling point.
5.3 💨 Sublimation and Deposition
- Sublimation: Some substances can change directly from a solid to a gas without becoming a liquid first. Example: Dry ice (solid carbon dioxide) or naphthalene balls.
- Deposition: This is the reverse, where a gas turns directly into a solid.
6. Evaporation
Evaporation is the process where a liquid turns into a vapor at any temperature below its boiling point. This is why a puddle of water disappears on a sunny day or wet clothes dry on a clothesline.
6.1 Factors Affecting Evaporation
Evaporation happens faster when:
- Surface Area is Increased: Spreading clothes out helps them dry faster.
- Temperature is Increased: Clothes dry faster on a hot day.
- Wind Speed is Increased: A windy day helps evaporate the water from clothes quickly.
- Humidity is Decreased: On a dry day (low humidity), the air can hold more water vapor, so evaporation is faster.
6.2 Evaporation Causes Cooling
When a liquid evaporates, it takes heat energy from its surroundings. This is why you feel cool when you sweat—the sweat evaporates from your skin, taking your body heat with it. It’s also why an earthen pot (matka) keeps water cool.
7. Extensive Practice Set
Here is a set of original questions to test your understanding of the chapter.
Part A: Multiple-Choice Questions (MCQs)
- Which of the following is NOT a characteristic of particles of matter?
a) They are in constant motion.
b) They are continuous and have no spaces.
c) They attract each other.
d) They have space between them. - When you dissolve sugar in water, the volume of water does not increase because the sugar particles:
a) Are very small.
b) Evaporate into the air.
c) Occupy the empty spaces between water particles.
d) Stick to the walls of the container. - The process where a gas turns directly into a solid is called:
a) Sublimation
b) Deposition
c) Condensation
d) Freezing - Which of the following will have the weakest force of attraction between its particles?
a) A piece of wood
b) Water
c) Oxygen gas
d) A sugar crystal - Why does a desert cooler work better on a hot, dry day?
a) Because the temperature is high and humidity is high.
b) Because the temperature is low and humidity is high.
c) Because the temperature is high and humidity is low.
d) Because the temperature is low and humidity is low.
Part B: Short Answer Questions
- Define “matter” and give two examples of things that are not matter.
- Why does the smell of hot food travel faster than the smell of cold food?
- What is the difference between boiling and evaporation?
- Give a reason why a gas is highly compressible, but a solid is not.
- What is latent heat of fusion?
Part C: Long Answer Questions
- Explain the three states of matter (solid, liquid, and gas) by comparing them on the basis of:
a) Shape and Volume
b) Force of attraction between particles
c) Kinetic energy of particles - Describe an activity to show that particles of matter are very small and are constantly moving. You can use the example of potassium permanganate or an incense stick to explain.
- Why do we feel cool after applying acetone (nail polish remover) to our palm? Explain the phenomenon responsible for this.
- Differentiate between sublimation and condensation with at least one example for each.
- What is the effect of changing temperature on a substance? Explain the process of melting and boiling with reference to the kinetic energy of particles and the forces of attraction between them.
Chapter Quiz
Also read: Class 9 Science Chapter 2 – Is Matter Around Us Pure?
