1. Introduction: Looking Inside the Atom
In the previous chapter, we learned about Dalton’s Atomic Theory, which stated that atoms are the fundamental, indivisible building blocks of matter. But this raised more questions: Are atoms really indivisible, or is there something smaller inside? Scientists at the end of the 19th century took up the challenge to look inside the atom, a story that begins with simple experiments on static electricity.
2. The Discovery of Subatomic Particles
Simple activities, like rubbing a comb on dry hair, show that objects can become electrically charged. This suggested that atoms must contain charged particles. This led to the discovery of three “subatomic” particles:
- ⚡ The Electron (e⁻): Discovered by J.J. Thomson, the electron is a negatively charged particle with a negligible mass.
- ➕ The Proton (p⁺): Discovered by E. Goldstein, the proton is a positively charged particle. Its mass is taken as one unit and is about 2000 times the mass of an electron.
- Neutron (n⁰): Discovered much later by J. Chadwick, the neutron has no charge and a mass nearly equal to that of a proton.
An atom is electrically neutral because the number of positive protons is balanced by the number of negative electrons. But how are these particles arranged?
3. Models of the Atom: How to Draw Something You Can’t See
3.1 Thomson’s “Plum Pudding” Model (1904)
J.J. Thomson proposed that an atom was a sphere of positive charge, with negatively charged electrons embedded in it, much like plums in a pudding. While it explained neutrality, it couldn’t explain the results of later experiments.
3.2 Rutherford’s Nuclear Model (1911)
Ernest Rutherford‘s team fired fast-moving, positively charged alpha (α)-particles at an extremely thin sheet of gold foil.
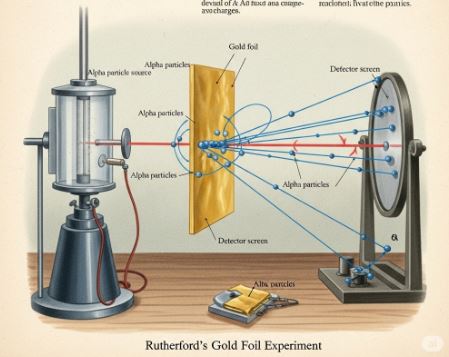
Rutherford’s experiment revealed that the atom is mostly empty space with a tiny, dense, positive nucleus.
The shocking results led to these conclusions:
- The atom is mostly empty space since most particles passed straight through.
- The atom has a tiny, dense, positively charged nucleus at its center where nearly all the mass resides.
- Electrons revolve around the nucleus in circular paths.
The Big Flaw: According to physics, a revolving electron should lose energy and spiral into the nucleus, making atoms unstable. But atoms are stable.
3.3 Bohr’s Model of the Atom (1913)
To fix Rutherford’s model, Niels Bohr proposed that electrons don’t just revolve anywhere. His key ideas were:
- Electrons can only revolve in certain special orbits called discrete orbits or energy levels (or shells).
- While in these orbits, electrons do not radiate energy, which explains why atoms are stable.
4. How Electrons Fill the Shells: The Bohr-Bury Rules
Bohr and Bury proposed rules for distributing electrons in energy shells (K, L, M, N…).
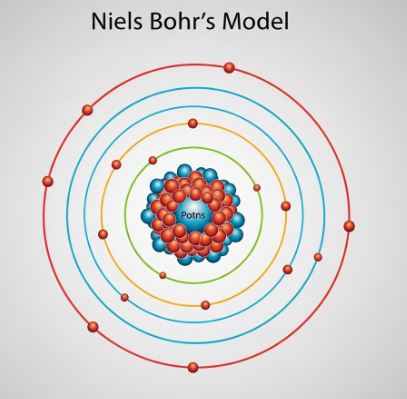
Bohr’s model confines electrons to specific energy shells, preventing them from collapsing into the nucleus.
- Rule 1 (The 2n² Rule): The maximum number of electrons in a shell is given by 2n², where ‘n’ is the shell number (e.g., K-shell (n=1) holds 2, L-shell (n=2) holds 8, M-shell (n=3) holds 18).
- Rule 2 (The Octet Rule): The outermost shell cannot hold more than 8 electrons.
- Rule 3 (Step-wise Filling): Electrons fill inner shells completely before entering outer shells.
5. Valency: An Atom’s Combining Power
The electrons in the outermost shell are called valence electrons. Atoms are most stable with a full outer shell, typically 8 electrons (an octet). Valency is how many electrons an atom must gain, lose, or share to achieve an octet.
- Example (Losing): Sodium (2, 8, 1) easily loses its 1 valence electron. Its valency is 1.
- Example (Gaining): Chlorine (2, 8, 7) easily gains 1 electron to complete its octet. Its valency is 1.
6. Atomic Number and Mass Number: The Atom’s ID
- ⚛️ Atomic Number (Z): The number of protons in the nucleus. This defines the element.
- 🔢 Mass Number (A): The total number of protons and neutrons in the nucleus.
We use the notation AZX to represent an atom. For example, Carbon is written as 126C.
7. Isotopes and Isobars: Atomic Variations
7.1 Isotopes
Isotopes are atoms of the same element with the same atomic number but different mass numbers (due to a different number of neutrons). For example, hydrogen has three isotopes: Protium (¹H), Deuterium (²H), and Tritium (³H). Since they have the same electron structure, isotopes have similar chemical properties.
7.2 Isobars
Isobars are atoms of different elements with different atomic numbers but the same mass number. For example, Argon (4018Ar) and Calcium (4020Ca) are isobars.
8. Extensive Practice Set
Part A: Multiple-Choice Questions (MCQs)
- Rutherford’s alpha-particle scattering experiment led to the conclusion that:
a) The atom is a solid positive sphere.
b) Electrons are embedded within a positive charge.
c) The mass and positive charge of an atom are concentrated in a small, dense nucleus.
d) Electrons revolve in fixed energy shells. - An atom has an atomic number of 13 and a mass number of 27. How many protons, electrons, and neutrons does this atom have?
a) 13 protons, 13 electrons, 14 neutrons
b) 13 protons, 14 electrons, 13 neutrons
c) 14 protons, 14 electrons, 13 neutrons
d) 13 protons, 13 electrons, 27 neutrons - Which of the following statements correctly describes isotopes?
a) Atoms of different elements with the same mass number.
b) Atoms of the same element with different numbers of protons.
c) Atoms of the same element with the same number of neutrons but different numbers of protons.
d) Atoms of the same element with different mass numbers. - According to the Bohr-Bury rules, what is the maximum number of electrons that can be accommodated in the M-shell (n=3)?
a) 2
b) 8
c) 18
d) 32 - The valency of an element with the electronic configuration 2, 8, 6 is:
a) 6
b) 8
c) 2
d) 1
Part B: Short Answer Questions
- Compare the key features of Thomson’s and Rutherford’s models of the atom. What was the major drawback of Rutherford’s model?
- Define Atomic Number (Z) and Mass Number (A). Write the complete symbol for an element that has 17 protons and 18 neutrons.
- The electronic configuration of a sodium atom is 2, 8, 1. How will it form a sodium ion (Na⁺)? What will be the electronic configuration of the ion?
- Name the three subatomic particles, and state their charge and location within an atom.
- What are isobars? Give one example.
Part C: Long Answer Questions
- Describe Niels Bohr’s model of the atom. How did his model overcome the major limitation of Rutherford’s model? Draw a simple diagram of Bohr’s model for an atom with three shells (K, L, M).
- An element has an atomic number of 15.
a) Write its electronic configuration.
b) How many valence electrons does it have?
c) What is its valency and why?
d) Is it a metal or a non-metal? - What are isotopes? Explain with the example of chlorine, which has two isotopes ³⁵Cl and ³⁷Cl. Why is the atomic mass of chlorine taken as 35.5 u?
- Explain the rules for the distribution of electrons in different energy shells as proposed by Bohr and Bury. Use these rules to show the electron distribution for Carbon (Z=6) and Argon (Z=18).
- A mystery element, X, has 11 protons and 12 neutrons. A second mystery element, Y, has 12 protons and 12 neutrons.
a) What are the atomic numbers and mass numbers of X and Y?
b) What is the relationship between X and Y? Are they isotopes? Why or why not?
c) What is the valency of element Y? Explain your reasoning.
Read Also:
Class 9 Is Matter Around Us Pure?
Chapter Quiz
Frequently Asked Questions (FAQs): Chapter 4- Structure of the Atom
1. What is Chapter 4- Structure of the Atom about?
Chapter 4- Structure of the Atom explains the internal structure of an atom, including the discovery of subatomic particles, atomic models proposed by scientists, electronic configuration, valency, atomic number, mass number, isotopes, and isobars. This chapter helps Class 9 students understand how atoms are built and how they behave.
2. Why is Chapter 4- Structure of the Atom important for Class 9 Science?
Chapter 4- Structure of the Atom is important because it forms the foundation of chemistry. Concepts like atomic number, mass number, valency, and electronic configuration are used in almost every chapter of chemistry in higher classes.
3. What are the main subatomic particles explained in Chapter 4- Structure of the Atom?
Chapter 4- Structure of the Atom explains three main subatomic particles: electrons (negative charge), protons (positive charge), and neutrons (no charge). Their discovery and properties help us understand atomic structure.
4. What is Rutherford’s model in Chapter 4- Structure of the Atom?
In Chapter 4- Structure of the Atom, Rutherford’s nuclear model states that an atom has a very small, dense, positively charged nucleus at its center, and electrons revolve around it. It proved that most of the atom is empty space.
5. Why was Bohr’s model introduced in Chapter 4- Structure of the Atom?
Bohr’s model was introduced in Chapter 4- Structure of the Atom to overcome the limitations of Rutherford’s model. It explained atomic stability by stating that electrons revolve in fixed energy levels without losing energy.
6. What are Bohr-Bury rules explained in Chapter 4- Structure of the Atom?
Chapter 4- Structure of the Atom explains Bohr-Bury rules for electron distribution. According to these rules, electrons fill shells step by step, the maximum electrons in a shell are given by 2n², and the outermost shell can have a maximum of 8 electrons.
7. What is valency according to Chapter 4- Structure of the Atom?
In Chapter 4- Structure of the Atom, valency is defined as the combining capacity of an atom. It depends on the number of electrons present in the outermost shell of an atom.
8. What is the difference between atomic number and mass number in Chapter 4- Structure of the Atom?
Chapter 4- Structure of the Atom defines atomic number as the number of protons in an atom, while mass number is the total number of protons and neutrons present in the nucleus.
9. What are isotopes explained in Chapter 4- Structure of the Atom?
Isotopes, as explained in Chapter 4- Structure of the Atom, are atoms of the same element that have the same atomic number but different mass numbers due to different numbers of neutrons.
10. What are isobars according to Chapter 4- Structure of the Atom?
According to Chapter 4- Structure of the Atom, isobars are atoms of different elements that have different atomic numbers but the same mass number.

