1. The Magic of Mixing: Understanding Mixtures
Hello students! Welcome to a fascinating journey into the world of chemistry that happens right inside your kitchen and all around you. Have you ever helped your parents by making lemonade on a hot summer day, or perhaps you’ve taken an Oral Rehydration Solution (ORS) when you were feeling unwell? Have you ever paused to wonder why the very first sip of your perfectly stirred homemade ORS tastes exactly the same as the very last sip? Why doesn’t it taste super salty at the beginning and purely sweet at the end?
The answer lies in the beautiful science of mixing! When we mix salt and sugar into water, these tiny particles spread out perfectly and evenly throughout the entire glass. Because the ingredients are distributed evenly from top to bottom, we call this a uniform mixture. But not everything mixes this beautifully. If you try to stir some chalk powder, sawdust, or dry sand into a glass of water, you will easily see the particles floating around or settling down heavily at the bottom of the container. Since the components are not evenly distributed, this type of mixture is known as a non-uniform mixture. Science is all about observing these small differences and understanding why they happen.
2. Who’s Who? Defining Solutes, Solvents, and Solutions
In our science classroom, we don’t just call perfectly mixed liquids “uniform mixtures”—we give them a special, scientific name. A uniform mixture is called a solution. Let’s break down the two main characters that make up every single solution you will ever encounter:
- The Solute: When you mix a solid into a liquid, the solid part that completely dissolves and disappears from plain sight is called the solute. Think of the sugar or salt you add to your water.
- The Solvent: The liquid part that does the heavy lifting of dissolving the solid is called the solvent. In most of our daily examples, water plays this crucial role beautifully.
So, the simple mathematical equation to remember in your mind is: Solute + Solvent = Solution.
But what if we mix two liquids together? How do we know which is the solute and which is the solvent? The rule is very simple: the substance that is present in the smaller amount is always the solute, and the one present in the larger amount is the solvent.
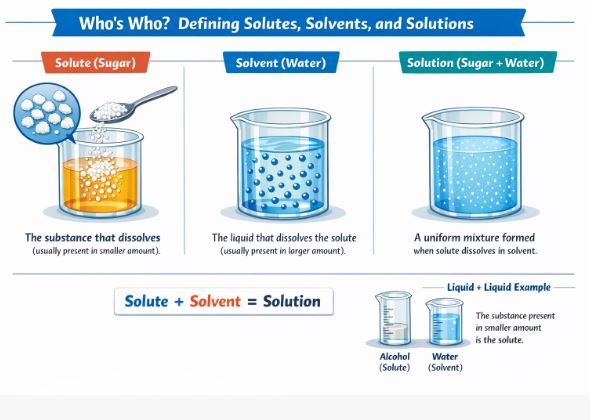
Figure-1: A solute evenly distributes into a solvent to create a uniform solution.
A Tasty Exception to Think About: Have you ever eaten the delicious Indian sweet, Gulab Jamun? The thick, sweet syrup (chashni) it swims in is made by dissolving a huge amount of solid sugar into a relatively small amount of liquid water. Even though there is physically more sugar than water, the water is still scientifically considered the solvent, and the sugar is the solute because it is the liquid dissolving the solid!
Are solutions only liquids? Not at all! Look around you right now. The invisible air you are breathing is actually a gaseous solution. Since nitrogen gas makes up the largest part of the air we breathe, we call nitrogen the solvent. Oxygen, carbon dioxide, argon, and other trace gases are present in much smaller amounts, so they are the solutes. Even solid solutions exist, like the brass or steel used to make your utensils!
3. The Capacity to Dissolve: Saturation, Concentration, and Solubility
Let’s do a fun mental experiment. Imagine you have half a glass of water, and you add one spoon of salt. You stir it vigorously, and it vanishes. You add another spoon, and it vanishes too. But what happens if you keep adding spoon after spoon after spoon? Eventually, no matter how hard you stir, the salt will stop dissolving and will just sit quietly at the bottom of your glass.
Why does this happen? Because the water has reached its maximum capacity! Once the water cannot dissolve any more solute at that specific room temperature, we say the solution has become a saturated solution. Before it hits this limit, while it can still happily accept and dissolve more salt, it is called an unsaturated solution.
The total amount of solute present in a specific amount of solvent is called its concentration. If you only add a tiny pinch of salt to a big jug of water, you have a dilute solution. If you add lots of salt, approaching that saturation limit, you have a concentrated solution. Furthermore, the absolute maximum amount of a solute that can successfully dissolve in a fixed quantity of solvent is known scientifically as its solubility.
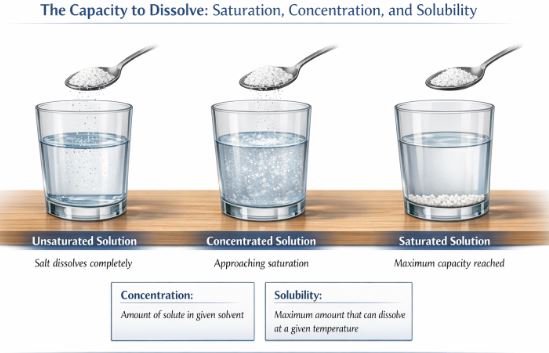
Figure-2: When a solvent reaches its limit, the extra solute settles at the bottom, creating a saturated solution.
4. Heating Things Up: How Temperature Affects Solubility
What if we want to force our saturated salt water to dissolve just a little bit more salt? Can we trick the water into doing more work? Yes, we can, by using the power of heat!
If you take a saturated solution (where the extra solid is just sitting at the bottom) and carefully heat it up on a laboratory stove to around 50°C or 70°C, you will notice something magical: the extra solid begins to dissolve completely! For most solid substances, solubility increases with an increase in temperature. Hot water has more internal “room” and energy to break apart and dissolve solids than cold water does. We can confidently say that a saturated solution at a normal temperature suddenly behaves like an unsaturated solution when you increase the temperature.
5. Do Gases Dissolve in Water? The Secret of Aquatic Life
We know solids like sugar and salt dissolve in water easily, but what about gases? Do they mix? Yes, they absolutely do, and life on Earth depends on it! Have you ever thought about how fish, aquatic plants, and other underwater organisms breathe? They survive by breathing the tiny, minute quantities of oxygen gas that have dissolved evenly into the water. Because the gases dissolve evenly, it forms a perfect, uniform mixture.
But here is a very important and surprising twist: while heating a liquid makes it better at dissolving solids, heating a liquid makes it worse at dissolving gases. The solubility of gases generally decreases as the temperature goes up. This means cold water holds much more oxygen than warm water. If a lake gets too hot during the intense summer months, it loses its dissolved oxygen, which can be very dangerous for the fish living in it, forcing them to move to deeper, cooler waters to survive.
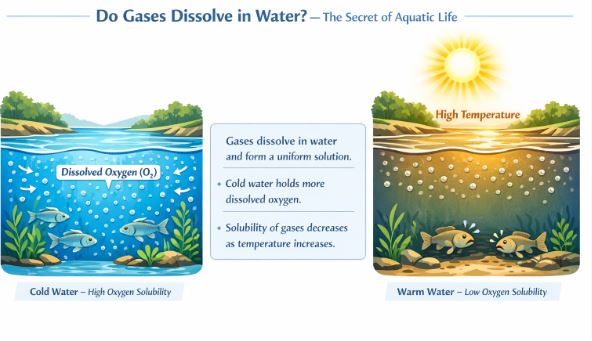
Figure-3: Aquatic creatures rely completely on the uniform solution of oxygen gas dissolved in water.
6. Float or Sink: Unlocking the Secret of Density
If you drop a heavy iron key and a wooden stick of the exact same size into a bucket of water, the key will sink instantly, but the wood will float happily on the surface. If you add oil to water, it floats right on top. Have you ever wondered why? We often loosely say the iron is “heavier,” but what we are really talking about is a special scientific property called density.
Think of density like a forest. If a forest has trees growing very close to each other, packed tightly, it is a dense forest. If the trees are far apart, it is less dense. In science, density is strictly defined as the mass present in a unit volume of a substance.
The mathematical formula you must remember is very simple:
Density = Mass / Volume
We measure mass in kilograms (kg) and volume in cubic metres (m3), so the standard SI unit for density is kg/m3. For smaller everyday liquids in the lab, we often use grams per millilitre (g/mL) or grams per cubic centimetre (g/cm3). Fun fact: The mass of 1 mL of pure water is almost exactly 1 gram at room temperature, making its density roughly 1 g/mL! If a block of aluminum has a density of 2.7 g/cm3, we say its relative density compared to water is 2.7, meaning it is 2.7 times denser than water.
7. How to Accurately Measure Volume and Density
To find the density of any object, we need to be careful detectives and measure two crucial things: its mass and its volume. We can measure mass very easily using a digital weighing balance. You place a watch glass on it, press the ‘tare’ or zero button to reset it, and then place your object on it to get the exact mass in grams. But how do we accurately measure volume?
For liquids, we use a tall, narrow, transparent glass container called a measuring cylinder. When you pour water into it, you will notice that the top surface of the water isn’t perfectly flat; it curves slightly inward. This curved surface is called the meniscus. To get a perfectly accurate reading for water or any colorless liquid, your eye must be exactly level with the bottom of this curve!

Figure-4: Always read the bottom of the meniscus at eye level for accurate liquid volume measurement.
What if you want to find the volume of an oddly shaped stone? You can’t use a geometry ruler for that! Instead, we use the genius water displacement method. You fill a measuring cylinder with some water and record the starting level. Then, you carefully tie a thread to the stone and gently lower it completely into the water. Naturally, the water level will rise. The exact difference between the new high water level and the original water level tells you the volume of the irregular stone!
8. How Temperature and Pressure Change Density
Did you know that you can actually change an object’s density without adding or removing any physical matter? Generally, when you heat a substance, its tiny internal particles gain energy, move faster, and spread further apart. This means the total volume expands and gets bigger, but the mass stays exactly the same. Since Density = Mass / Volume, having a larger volume on the bottom of the equation means the overall density decreases.
This is exactly the science of how giant hot air balloons fly! By heating the air inside the giant balloon canopy, the air expands and becomes significantly less dense than the cool, heavy air outside. This causes the massive balloon to rise gracefully into the sky.
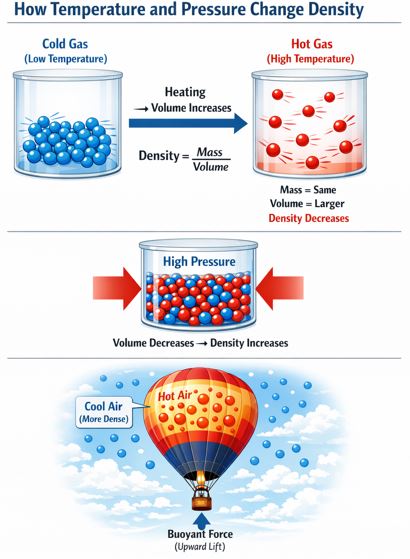
Figure-5: Hot air is less dense than cool air, creating an upward buoyant force that allows balloons to fly.
Pressure also plays a role, especially for gases. If you apply high pressure to a gas, you squish the particles closer together, decreasing the volume and increasing the density. Liquids and solids are barely affected by pressure because their particles are already packed so tightly together.
The Great Ice Exception: Here is a spectacular twist of nature. Almost all solids are denser than their liquid forms. But water is incredibly special. When water temperature drops and turns into solid ice at 0°C, the water particles arrange themselves in a unique, structured pattern that actually takes up more space. Because this same amount of water now occupies a larger volume, its density decreases. As a result, ice becomes lighter than liquid water and floats on its surface. This amazing property creates a floating thermal blanket of ice on cold winter lakes, keeping the water underneath warm enough for fish and aquatic creatures to survive extreme cold weather!
9. Digging Deeper: Earth’s Layers and Ancient Traditions
Density isn’t just about water and balloons; it’s about the entire planet! Our Earth is composed of several layers: the crust, upper mantle, lower mantle, outer core, and inner core. The outermost layer, the crust we live on, is the lightest and least dense. As you travel deeper towards the center of the Earth, extreme pressure and temperature make the materials heavier, highly compact, and incredibly dense.
We also see practical uses of density in human history. In ancient times, before large metal ships were engineered, people used bamboo poles and wooden logs to travel across rivers. Bamboo was an incredible choice because it is naturally hollow, very light, and has a much lower density than water, meaning it floats easily. People tied these together to make simple rafts for fishing and trading, a practice still seen today in some regions.
Furthermore, understanding solutions is deeply rooted in our traditions. In Ningel village in Manipur, people still produce salt using traditional methods by drawing up highly concentrated salty water from deep wells. They boil this solution over firewood until all the solvent (water) evaporates, leaving behind pure solute (salt crystals) which they shape into traditional ‘salt cakes’. This is a beautiful, real-world application of evaporating a solvent to recover a solute!
10. Common Student Misconceptions (Teacher’s Note!)
- Misconception: “Melting and dissolving are the exact same thing.”
Correction: They are very different! Melting requires heat to physically turn a solid into a liquid state (like an ice cube melting into water). Dissolving is mixing a solid into a separate liquid solvent to form a uniform mixture (like stirring sugar into water). - Misconception: “Heavy objects always sink no matter what.”
Correction: It’s not about the total weight or mass; it’s entirely about density. A massive, incredibly heavy steel cruise ship can float beautifully because its shape contains a massive volume of air inside, making its overall average density lower than water. A tiny, lightweight solid pebble sinks instantly because its density is tightly packed and higher than water. - Misconception: “Gases can’t dissolve in liquids; only solids can.”
Correction: They definitely can! Aquatic life depends completely on dissolved oxygen gas. Also, the refreshing fizz in your cold soda is simply dissolved carbon dioxide gas.
Practice Questions (CBSE Pattern)
A. Very Short Answer Questions
Q1: What is a solution?
Answer: A solution is a uniform mixture formed when two or more substances mix completely and evenly, such that the components cannot be seen separately.
Q2: Identify the solvent and the major solutes present in the air around us.
Answer: In the air, nitrogen gas is present in the largest amount and is considered the solvent. Oxygen, carbon dioxide, argon, and other gases are present in smaller amounts and are considered the solutes.
Q3: Write the mathematical formula for density.
Answer: Density is calculated as Mass divided by Volume (Density = Mass / Volume).
B. Short Answer Questions
Q1: What is the main difference between a saturated solution and an unsaturated solution?
Answer: An unsaturated solution is one where more solute can still be dissolved at a given temperature. A saturated solution is one that has reached its maximum capacity; it cannot dissolve any more solute at that specific temperature, and any extra solid will simply settle at the bottom undissolved.
Q2: Why do fish and aquatic animals survive better in cold water compared to hot water?
Answer: Aquatic animals rely on oxygen gas dissolved in the water. The solubility of gases generally decreases as the temperature increases. Therefore, cold water can hold much more dissolved oxygen than hot water, ensuring sufficient oxygen for the survival of aquatic life.
Q3: Why do ice cubes float on liquid water instead of sinking to the bottom?
Answer: When water cools down and freezes into solid ice at 0°C, its particles arrange in a unique structure that actually takes up more space, a process called expansion. Because the same mass of water now occupies a larger volume, the density of ice becomes less than that of liquid water, causing it to be lighter and float on the surface.
C. Long Answer Questions
Q1: Describe step-by-step how you would calculate the density of a small, irregularly shaped stone using laboratory equipment.
Answer:
1. First, measure the exact mass of the dry stone. Place a clean watch glass on a digital weighing balance, press the tare (reset) button to bring it to zero, and place the stone on it. Record the mass in grams.
2. Next, measure the volume using the water displacement method. Take a measuring cylinder and fill it partially with water. Record this initial volume reading carefully at eye level with the bottom of the meniscus.
3. Tie the stone with a thin thread and gently lower it completely into the water inside the measuring cylinder.
4. Record the new, higher final volume reading.
5. Subtract the initial volume from the final volume to find the volume of the displaced water. This difference is exactly equal to the volume of the stone.
6. Finally, calculate the density using the formula: Density = Mass / Volume.
Q2: How does changing the temperature affect the density of substances, and how does a hot air balloon utilize this scientific principle to fly?
Answer: Generally, when a substance is heated, the thermal energy causes its particles to move away from each other and spread out. This causes the physical volume of the substance to increase, while the actual mass remains exactly the same. Since density is mass divided by volume, an increase in volume results in a decrease in density upon heating.
A hot air balloon works perfectly on this principle. A large burner heats the air trapped inside the balloon envelope. As this air heats up, it expands and its volume increases, making the hot air inside significantly less dense than the cooler air surrounding it outside. Because objects with lower density rise above fluids with higher density, the less dense hot air creates a buoyant force that lifts the entire balloon high into the sky. Conversely, when cooling down, volume decreases and density increases.
D. Case-Based Question
Rahul is conducting an experiment in his kitchen. He takes a glass containing exactly 100 mL of water at normal room temperature and keeps adding spoons of salt to it while stirring continuously. After successfully dissolving 5 large spoons of salt to form a uniform mixture, he notices that when he adds the 6th spoon of salt, it simply refuses to dissolve and settles entirely at the bottom of the glass, no matter how hard he stirs.
Q1: What specific scientific term describes the state of Rahul’s solution after the 6th spoon is added and settles at the bottom?
Answer: The solution has become a saturated solution at that specific room temperature, meaning it cannot dissolve any more solute.
Q2: If Rahul desperately wants to dissolve that 6th spoon of salt without adding any more liquid water, what physical change should he apply to the glass container?
Answer: He should heat the solution on a stove. Increasing the temperature generally increases the solubility of solid solutes in liquid solvents, which will allow the undissolved salt to completely dissolve.
E. Assertion–Reason
Assertion (A): When you accidentally pour cooking oil into a glass filled with water, the oil will immediately separate and float on the top surface of the water.
Reason (R): The physical density of the cooking oil is significantly lower than the density of the water.
Answer: Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct scientific explanation for Assertion (A). In fluid dynamics, liquids or objects that possess a lower density will naturally float on top of liquids that possess a higher density.
Read Also
For official syllabus and textbooks, visit the
NCERT Official Website.

