1. Introduction: What is Stuff Made Of? |
Hello students! Have you ever looked at a piece of chalk, a drop of water, or the smoke rising from an incense stick and wondered, “What is this actually made of?”
In our daily lives, we see continuous objects. A wooden table looks like one solid block. Water looks like a continuous stream. But if we had magic glasses that could zoom in millions of times, what would we see? We would see that everything around us—matter—is not continuous like a sheet of paper. Instead, it is made up of tiny, tiny pieces. We call these pieces particles.
Think of a sand castle. From far away, it looks like a smooth structure. But when you go close, you see it is made of millions of tiny grains of sand. Matter is very similar, but the particles are much, much smaller than sand grains!
2. Breaking It Down: The Chalk Experiment
Let’s perform a thought experiment. Imagine you take a stick of chalk.
1. Break it in half.
2. Break that half into smaller pieces.
3. Crush those pieces into a coarse powder.
4. Grind that powder into a fine dust.
Is the fine dust still chalk? Yes! It has the same white colour and properties. If we could keep breaking it down further, beyond what our eyes can see, we would eventually reach the smallest unit of chalk. These are the constituent particles. This tells us that matter is particulate (made of particles), not continuous.
3. The Mystery of Disappearing Sugar
One of the best ways to prove that matter has “empty spaces” inside it is the sugar-water experiment. If you take a glass full of water and add a spoon of sugar, stir it well, the sugar disappears. We say it has dissolved.
But here is the interesting question: Does the water level rise?
If you measure carefully, you will notice the water level barely changes. Why? If we added something to the water, the volume should go up, right?
The answer lies in the Interparticle Space. Water is made of particles that have small gaps (spaces) between them. When sugar dissolves, the tiny sugar particles break apart and hide in these empty spaces between the water particles. This confirms two things:
- Matter is made of particles.
- There is space between these particles.
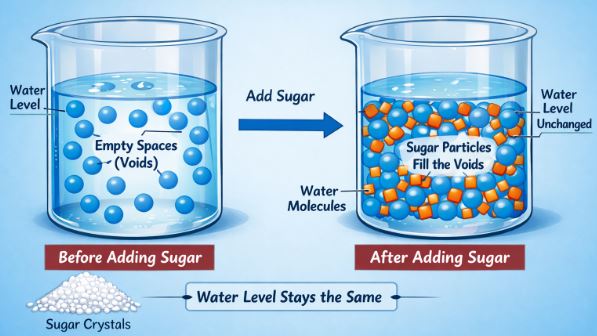
Figure-1: When sugar dissolves, its particles occupy the empty spaces (voids) between water particles, so the water level remains mostly unchanged.
4. The Invisible Force: What Holds Matter Together?
If everything is made of loose particles, why doesn’t a wooden chair fall apart into dust? Why does a drop of water stay together?
There is a special glue holding these particles together. In science, we don’t call it glue; we call it the Force of Attraction. The particles attract each other.
- Strong Force: In a steel spoon, the particles hold each other very tightly. You cannot break it easily.
- Weak Force: In a piece of chalk, the force is weaker, so you can break it.
- Very Weak Force: In water, you can easily move your hand through it because the force is weak enough to let particles move apart temporarily.
5. The Three States of Matter
Depending on how much space is between particles and how strong the force of attraction is, matter exists in three main forms (states): Solids, Liquids, and Gases.
A. The Solid State
Think of a class of disciplined students sitting in their assigned rows. They can wiggle in their seats, but they cannot leave their place. This is a solid.
- Packing: Particles are tightly packed.
- Space: Minimum interparticle space.
- Force: Maximum force of attraction.
- Movement: Particles cannot move freely; they only vibrate in their fixed positions.
- Result: Solids have a fixed shape and a fixed volume. They are rigid.
B. The Liquid State
Now, imagine students during a lunch break inside the classroom. They are still inside the room (volume is fixed), but they are moving around, chatting, and mingling. They are not stuck to one desk. This is a liquid.
- Packing: Loosely packed compared to solids.
- Space: Moderate interparticle space.
- Force: Weaker attraction than solids (allows flow).
- Movement: Particles can slide past one another.
- Result: Liquids have a fixed volume but NO fixed shape. They take the shape of the container.
C. The Gaseous State
Finally, imagine the school bell rings and students rush out to the playground. They run everywhere, far apart from each other. This is a gas.
- Packing: Very loosely packed.
- Space: Maximum interparticle space.
- Force: Negligible (almost zero) attraction.
- Movement: Particles move randomly at high speeds in all directions.
- Result: Gases have NO fixed shape and NO fixed volume. They fill the entire container.
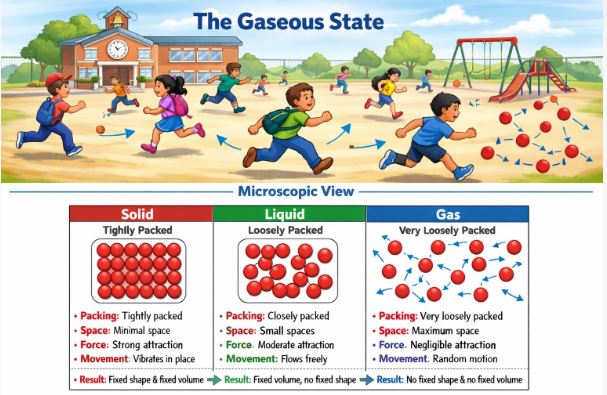
Figure-2: A microscopic view showing how particles are arranged in Solids (tight), Liquids (loose), and Gases (free).
6. Compressibility: The Syringe Activity
Can we squeeze matter? It depends on the state. Let’s look at the “Syringe Activity” mentioned in your curriculum.
If you take a syringe (without the needle), close the nozzle with your thumb, and try to push the piston:
- With Chalk Powder (Solid): You cannot push the piston. Solids are not compressible.
- With Water (Liquid): You can barely push it. Liquids are almost incompressible.
- With Air (Gas): You can push the piston down quite a bit!
Why? Because gases have huge empty spaces between particles. When we push, we are just forcing the particles to come closer into those empty spaces. Solids and liquids barely have any space left to squeeze into.
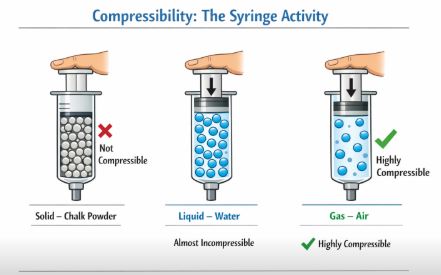
Figure-3: We can compress gases easily because there is a lot of empty space between their particles.
7. How Particles Move: Diffusion and Temperature
Particles are never completely still. They are always moving or vibrating. This movement is linked to Temperature (Heat Energy).
The Incense Stick Example:
If you light an incense stick in one corner of the room, you can smell it in the opposite corner within seconds. How? The particles of the smoke mix with the air particles and drift across the room. This mixing of particles on their own is called Diffusion.
Hot vs. Cold Water:
If you drop a crystal of Potassium Permanganate (purple colour) into hot water and another into cold water, what happens?
The purple colour spreads much faster in the hot water.
Conclusion: When temperature increases, the particles gain energy and move faster. Heat increases the kinetic energy of particles.
8. Changing States: Melting and Boiling
We can change the state of matter by changing the temperature.
- Solid to Liquid (Melting): When we heat a solid (like ice), the particles start vibrating faster. Eventually, they vibrate so hard that they overcome the strong force holding them. They break free and start moving around. The solid melts into a liquid.
- Liquid to Gas (Boiling/Evaporation): If we heat the liquid further, the particles move even faster. They eventually have enough energy to completely break away from each other and fly off. The liquid becomes a gas.
9. Historical Insight: Our Indian Roots
Did you know? Thousands of years before modern microscopes, an Indian philosopher named Acharya Kanad proposed this theory. He said if we keep dividing matter, we will get smaller and smaller particles, and ultimately, we will find a particle that cannot be divided further. He called this “Parmanu” (Atom).

Figure-4: Ancient Indian philosopher Acharya Kanad gave the concept of ‘Parmanu’ long before modern science.
10. Common Misconceptions to Avoid
Students often get confused about these points. Let’s clear them up:
- Misconception: “Sand takes the shape of a jar, so is sand a liquid?”
Fact: No! Sand is a solid. Each individual grain of sand has a fixed shape. A heap of sand can change shape, but the particle (grain) does not. - Misconception: “When sugar dissolves, it disappears/vanishes.”
Fact: It does not vanish. It is still there (you can taste the sweetness). The particles just became too small to see and hid in the gaps of water. - Misconception: “Air weighs nothing.”
Fact: Air is matter. It has mass and occupies space. An inflated balloon is heavier than a deflated one!
Practice Questions (CBSE Pattern)
A. Very Short Answer Questions
Q1: What is the smallest unit of matter called in general terms for this chapter?
Answer: Constituent particles.
Q2: Which state of matter has the strongest force of attraction between particles?
Answer: Solid state.
Q3: Define melting point.
Answer: The temperature at which a solid melts to become a liquid at atmospheric pressure.
B. Short Answer Questions
Q1: Why can we wave our hand through air easily, but not through a wooden block?
Answer: In air (gas), particles are far apart with negligible force of attraction, allowing easy movement. In a wooden block (solid), particles are tightly packed with very strong forces, preventing our hand from passing through.
Q2: Explain why the smell of hot food reaches us faster than cold food.
Answer: Particles move faster at higher temperatures due to increased kinetic energy. Therefore, the aromatic particles of hot food diffuse into the air and reach us quickly, unlike cold food.
Q3: Sponge is a solid, yet we can compress it. Why?
Answer: A sponge has many minute holes (pores) in which air is trapped. When we press it, the air is expelled out, allowing it to compress. The material itself is still solid.
Q4: Why do liquids have a fixed volume but not a fixed shape?
Answer: The attraction force in liquids is strong enough to keep particles together (fixed volume) but not strong enough to hold them in a rigid position, so they slide past each other to take the container’s shape.
C. Long Answer Questions
Q1: Describe an activity to show that particles of matter have space between them.
Answer:
1. Take a glass beaker filled halfway with water. Mark the water level.
2. Add two spoons of sugar or salt.
3. Stir until it dissolves completely.
4. Observe the water level again.
Observation: The water level does not rise significantly.
Conclusion: The sugar particles broke down and occupied the empty spaces (voids) between the water particles, proving that space exists between particles of matter.
Q2: Differentiate between Solids, Liquids, and Gases based on: (a) Interparticle Distance, (b) Compressibility, (c) Particle Movement.
Answer:
(a) Interparticle Distance: Minimum in solids, moderate in liquids, maximum in gases.
(b) Compressibility: Negligible in solids and liquids, very high in gases.
(c) Particle Movement: Particles only vibrate in solids; they slide/flow in liquids; they move freely and randomly in gases.
D. Case-Based Question
Case: Rahul inflated a balloon and tied it. Then he tried to push the balloon inwards and found he could change its shape easily. Next, he took a glass marble and tried to change its shape by pressing it but failed.
Q1: Why could Rahul compress the balloon?
Answer: The balloon contains air (gas), which has large spaces between particles, making it compressible.
Q2: What does the marble representing in terms of particle arrangement?
Answer: The marble is a solid where particles are tightly packed with no space to move, making it rigid and incompressible.
E. Assertion–Reason
Assertion: A gas fills the entire vessel in which it is kept.
Reason: The force of attraction between gas particles is negligible, and they move freely in all directions.
Answer: Both Assertion and Reason are correct, and the Reason is the correct explanation for the Assertion.
Read Also
For official syllabus and textbooks, visit the
NCERT Official Website.

