Introduction | Class 11 Biology Chapter 12 Respiration in Plants notes
Hello students! Welcome back to another exciting journey into the microscopic world of biology. In our previous classes, we talked extensively about how plants make their food using sunlight—a magical process we call photosynthesis. But let me ask you a simple question: What happens after the food is made?
Think about your own daily life. You eat a hearty breakfast to get energy for school, sports, and studying. But just having a sandwich sitting in your stomach doesn’t give you energy. Your body has to break it down, extract the energy locked inside the food molecules, and convert it into a usable form. Plants, animals, and even tiny bacteria face the exact same challenge!
In this chapter, we are going to explore Cellular Respiration. This is the fascinating biological machinery that takes the food (like glucose), breaks its chemical bonds step-by-step, and releases energy. This energy is then trapped in small, usable packets called ATP (Adenosine Triphosphate), which acts as the ‘energy currency’ of the cell. Whenever a cell needs to do some work, it simply spends some ATP. Let’s dive in and see how this amazing energy-extraction process works!
1. Do Plants Actually Breathe?
When we think of breathing, we think of lungs, noses, and chests expanding. So, do plants breathe? Well, they don’t have a specialized respiratory system like we do. They don’t have lungs or a heart to pump oxygen around. But yes, they absolutely need oxygen to survive, and they do release carbon dioxide.
How do they manage without lungs? Nature has given them a very simple and decentralized system. Here is why plants can get away without having complex breathing organs:
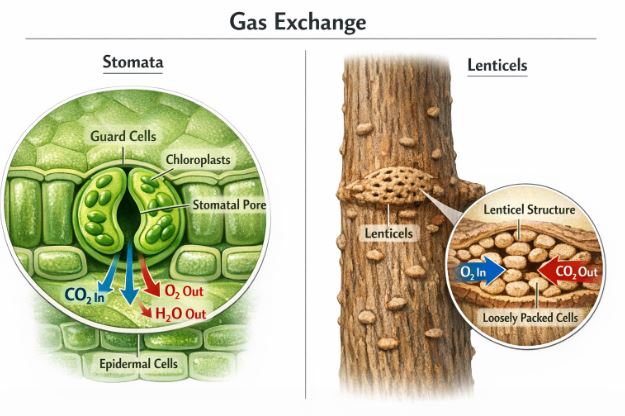
- Every part looks after itself: In a plant, the roots, stems, and leaves take care of their own gas exchange. There is very little transport of gases from one part of the plant to another. The leaves have tiny pores called stomata, and the woody stems have small openings called lenticels to let air in and out.
- Low energy demands: Plants don’t run around, hunt, or fly. Therefore, their roots, stems, and leaves respire at rates far lower than animals do. They just don’t need a massive, continuous rush of oxygen.
- Internal oxygen supply: During the day, leaves are actively performing photosynthesis, which produces oxygen right inside the cells! So, the cells have an in-house supply of oxygen precisely when they are most active.
- Short diffusion distances: Even in a massive, thick banyan tree, the living cells are located close to the surface (just beneath the bark). The core of the thick trunk is actually dead wood, which only provides mechanical support and doesn’t need oxygen. Furthermore, the loose arrangement of cells (parenchyma) in plants creates a network of air spaces, ensuring that almost every living cell is in direct contact with air.
2. Glycolysis: The Universal First Step
Whether you are a human, a mango tree, or a bacteria living in the mud, if you are breaking down glucose for energy, you start here.
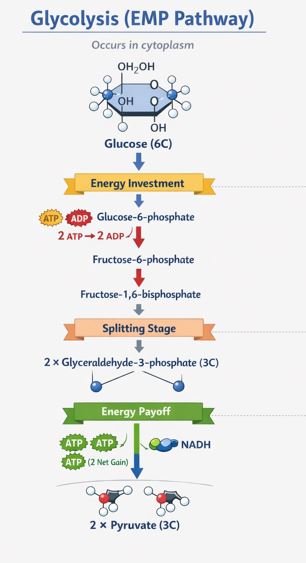
The term Glycolysis comes from two Greek words: glycos meaning sugar, and lysis meaning splitting. Quite literally, it is the splitting of sugar. This entire process happens in the cytoplasm of the cell. Importantly, glycolysis does NOT require oxygen. It is an anaerobic process, making it the only source of energy for completely anaerobic organisms.
Scientists Embden, Meyerhof, and Parnas mapped out this pathway, which is why it is frequently called the EMP pathway.
The 10-Step Journey (Simplifying the Chemistry)
Imagine glucose, a stable 6-carbon molecule, as a tough piece of wood. You can’t just snap it instantly to get energy. You have to weaken it first.
Phase 1: The Investment Phase (Spending Energy)
To start the breakdown, the cell actually has to spend some energy. It uses 2 ATP molecules to add phosphate groups to the glucose, eventually turning it into a highly unstable molecule called Fructose-1,6-bisphosphate. Because it is so unstable, it immediately snaps perfectly in half! You now have two 3-carbon molecules (like PGAL).
Phase 2: The Payoff Phase (Gaining Energy)
Now, the cell gets its return on investment. These two 3-carbon molecules undergo a series of chemical changes. During these steps:
- Hydrogen atoms are extracted and given to a carrier molecule called NAD+, turning it into NADH + H+. Think of NADH as a high-value cheque that the cell can cash in later for a lot of ATP.
- Phosphate groups are plucked off and attached to ADP to create fresh, usable ATP.
The Final Result: At the end of the 10 steps, the original 6-carbon glucose has been converted into two molecules of a 3-carbon compound called Pyruvic Acid (or Pyruvate).
Teacher’s Tip on the Math: We spent 2 ATPs at the start. We produced 4 ATPs by the end. Therefore, the net direct gain of glycolysis is exactly 2 ATP molecules per glucose, plus 2 molecules of the energy-rich NADH.
3. The Crossroads: What happens to Pyruvic Acid?
Now the cell has Pyruvic acid. What it does next completely depends on one crucial factor: Is oxygen available?
If oxygen is absent (anaerobic conditions), the cell undergoes Fermentation. If oxygen is present (aerobic conditions), it proceeds to Aerobic Respiration. Let’s look at life without oxygen first.
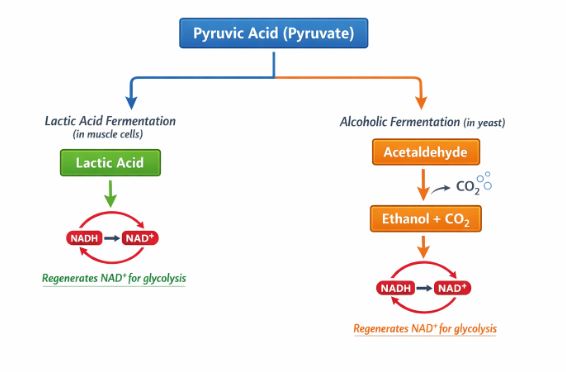
3.1 Fermentation (Anaerobic Respiration)
When there is no oxygen to help extract the massive amount of energy left in pyruvic acid, the cell goes into survival mode. It performs incomplete oxidation. The main goal here isn’t really to make tons of energy; the main goal is to recycle the NAD+ carriers so that glycolysis (which gives a tiny 2 ATP) can at least keep running! There are two main types:
-
Alcoholic Fermentation: Occurs in organisms like yeast. The pyruvic acid is converted into ethyl alcohol (ethanol) and carbon dioxide gas. The enzymes involved are pyruvic acid decarboxylase and alcohol dehydrogenase.
Real-world connection: This is why bread dough rises! The yeast ferments the sugar, and the released CO2 gas bubbles get trapped in the dough, making it fluffy. However, this process is toxic to the yeast itself. If the alcohol concentration reaches about 13%, the yeast poisons itself and dies! - Lactic Acid Fermentation: Occurs in certain bacteria (like the ones that turn milk into curd) and, surprisingly, in your own body! When you do heavy exercise, your muscles need energy faster than your blood can deliver oxygen. Your muscle cells temporarily switch to lactic acid fermentation. The buildup of lactic acid is what causes those painful muscle cramps after a heavy workout.
The Verdict on Fermentation: It is highly inefficient. Less than 7% of the energy trapped in glucose is released, and it produces hazardous byproducts like alcohol or acid.
4. Aerobic Respiration: The Ultimate Powerhouse
If oxygen is present, the cell can extract an enormous amount of energy. The pyruvic acid packs its bags and leaves the cytoplasm to enter the VIP section of the cell: the Mitochondria. Aerobic respiration completely breaks down the organic molecules into CO2 and water, releasing massive amounts of ATP.
4.1 The Gateway Step (Link Reaction)
Before pyruvic acid (3-carbon) can enter the main cycle, it gets prepped in the mitochondrial matrix. It undergoes oxidative decarboxylation. Simply put, it loses a carbon (which flies off as CO2 gas) and gives off some hydrogen to form NADH. The remaining 2-carbon fragment attaches to a carrier called Coenzyme A to form Acetyl CoA. Acetyl CoA is the golden ticket to enter the next phase.
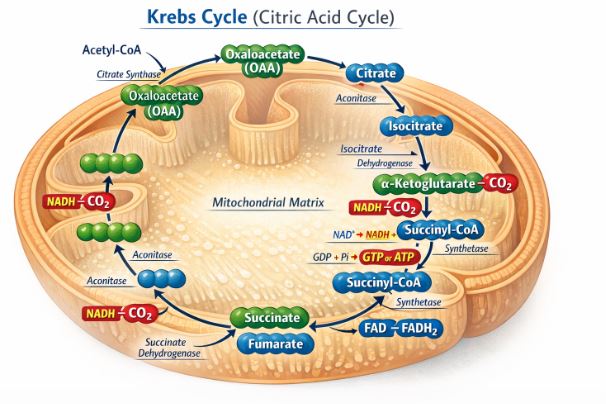
4.2 The Tricarboxylic Acid (TCA) Cycle / Krebs Cycle
Named after the brilliant scientist Hans Krebs, this is a continuous biochemical merry-go-round located in the mitochondrial matrix.
1. The 2-carbon Acetyl CoA enters the cycle and joins hands with a 4-carbon molecule called Oxaloacetic acid (OAA) to form a 6-carbon molecule called Citric Acid. Because citric acid is the first product, this is also called the Citric Acid Cycle.
2. As the cycle turns, the molecule undergoes a series of transformations. Carbon is progressively ripped off and released as CO2 (this is the very CO2 that you exhale!).
3. More importantly, at multiple points in the cycle, high-energy hydrogen atoms are harvested. They are picked up by our carrier molecules, creating 3 molecules of NADH and 1 molecule of a similar carrier called FADH2 per turn.
4. One molecule of ATP (in the form of GTP) is also generated directly during the cycle.
5. Finally, the molecule is converted back into the 4-carbon OAA, ready to accept another Acetyl CoA and keep the cycle spinning.
Remember: Since one glucose molecule gave us TWO pyruvic acids, the Krebs cycle turns TWICE for every molecule of glucose.
4.3 The Electron Transport System (ETS) and Oxidative Phosphorylation
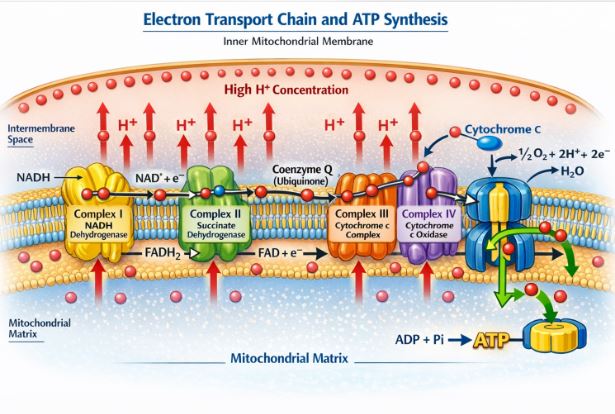
So far, we have only directly made a tiny bit of ATP. But we have a huge pile of “cheques” in the form of NADH and FADH2. It’s time to go to the bank and cash them in! This happens on the inner mitochondrial membrane, which is packed with a series of protein complexes known as the Electron Transport System (ETS).
How the machinery works:
- The NADH and FADH2 drop off their high-energy electrons at the start of the ETS chain.
- These electrons are passed like a hot potato from one protein complex (Complex I, II, III, IV) to another down the chain.
- As the electrons jump, they release energy. The membrane proteins use this energy to actively pump Protons (H+ ions) out of the matrix and into the inter-membrane space. This creates a massive concentration gradient—like pumping water up into a high dam.
- The Final Acceptor: At the very end of the chain, the electrons are exhausted. Who catches them? Oxygen! Oxygen acts as the ultimate garbage collector. It grabs the electrons, picks up a couple of protons, and turns into harmless metabolic water (H2O). Without oxygen sitting at the end to catch the electrons, the entire chain would get blocked up, and the whole system would shut down.
Making the ATP (Complex V):
Now we have a huge buildup of protons (H+) trapped in the inter-membrane space. They desperately want to flow back into the matrix. The only way back is through a special turbine-like enzyme called ATP Synthase (Complex V). As the protons rush through this channel (like water rushing out of a dam), the enzyme spins and physically jams an ADP and a Phosphate together to create ATP!
Every NADH that cashes in its electrons provides enough pumping power to generate roughly 3 ATPs. Every FADH2 generates about 2 ATPs. Because this ATP synthesis relies on the oxidation of these carriers by oxygen, the process is called Oxidative Phosphorylation.
5. The Respiratory Balance Sheet
Let’s do some accounting. If we add up the 2 net ATPs from glycolysis, the 2 direct ATPs from the Krebs cycle, and cash in all the NADH and FADH2 molecules at the ETS bank, we get a grand total of up to 38 ATP molecules from a single molecule of glucose! Compare that to the measly 2 ATPs produced by fermentation. Aerobic respiration is incredibly efficient.
However, we must understand that this 38 ATP figure is highly theoretical. It relies on strict assumptions:
- It assumes the pathways work in a perfect, orderly sequence (Glycolysis → Link → Krebs → ETS) without interruptions.
- It assumes no intermediate molecules are pulled out to build other things.
- It assumes only pure glucose is being respired.
In a living, dynamic cell, these assumptions aren’t entirely true. Pathways work simultaneously, molecules are constantly pulled in and out, and the cell uses whatever fuel is available. Still, calculating this gives us a great appreciation for the sheer efficiency of cellular machinery.
6. The Amphibolic Pathway: A Two-Way Street
Traditionally, respiration is taught as a catabolic process. Catabolism means breaking things down (like a demolition crew breaking down a building into bricks). Glucose is demolished into CO2 and water to get energy.
But nature is highly resourceful. The cell doesn’t just use these pathways for demolition. If the cell suddenly needs to build a fat molecule or a protein, it doesn’t start from scratch. It dips its hand into the respiratory pathway, pulls out an intermediate molecule (like Acetyl CoA), and diverts it to a building process! Building molecules is called anabolism.
Because the respiratory pathway is heavily involved in BOTH breaking down fuels for energy (catabolism) and providing the building blocks to synthesize new molecules (anabolism), scientists correctly refer to it as an Amphibolic Pathway (“Amphi” meaning both).
7. Respiratory Quotient (RQ)
Different foods burn differently. The Respiratory Quotient (RQ) is a simple mathematical ratio that helps us understand exactly what kind of fuel a plant or animal is currently burning for energy.
RQ = (Volume of CO2 evolved) / (Volume of O2 consumed)
- Carbohydrates (RQ = 1): When sugars are burned aerobically, they require exactly the same amount of oxygen molecules as the carbon dioxide molecules they produce. (e.g., 6 CO2 / 6 O2 = 1).
- Fats (RQ < 1): Fats are massive, complex molecules that have very little inherent oxygen in their structure. To break them down completely, the cell has to consume a massive amount of external oxygen compared to the CO2 released. For a fat like tripalmitin, the RQ is around 0.7.
- Proteins: If proteins are forced to be used as fuel, their RQ sits around 0.9.
Teacher’s Note: In healthy living organisms, pure proteins or fats are rarely used alone. The cell burns a mixture of substrates, so the overall RQ is usually a blend.
Real-Life Examples to Understand Respiration
- The Bread Dough Magic: Ever wondered why bread has tiny holes in it? When baker’s yeast is mixed into the dough, it is buried away from oxygen. It undergoes alcoholic fermentation. The carbon dioxide gas it produces gets trapped in the sticky gluten of the dough, forcing it to rise and creating the fluffy texture we love. When baked, the alcohol evaporates safely away!
- The “Burn” of a Sprint: During a relaxed walk, your muscles get plenty of oxygen and perform aerobic respiration (yielding 38 ATP). But if a dog chases you and you sprint at top speed, your heart cannot pump oxygen fast enough. Your muscle cells panic and switch to lactic acid fermentation to get quick (though inefficient) energy. The toxic buildup of lactic acid is the burning sensation and cramps you feel afterward. Heavy breathing post-run is your body taking in extra oxygen to clear out that lactic acid debt.
Key Takeaways & Summary
- Cellular respiration is the breakdown of complex organic molecules (mostly glucose) to release trapped energy, storing it as ATP.
- Plants lack lungs but exchange gases via stomata, lenticels, and loosely packed cells.
- Glycolysis (Cytoplasm) splits one 6C glucose into two 3C pyruvic acids, yielding a net of 2 ATP and 2 NADH. It requires no oxygen.
- In the absence of oxygen, Fermentation occurs (producing lactic acid in muscles, or ethanol + CO2 in yeast). It’s very inefficient.
- In the presence of oxygen, Pyruvic acid enters the mitochondria, converts to Acetyl CoA, and enters the Krebs Cycle, completely oxidizing into CO2 while filling carrier molecules (NADH/FADH2).
- The ETS on the inner mitochondrial membrane cashes in these carriers to pump protons. ATP Synthase uses this proton flow to generate massive amounts of ATP. Oxygen acts as the final electron catcher, forming water.
- The respiratory pathway is amphibolic, serving as both a breakdown route and a source of building blocks.
- RQ is the ratio of CO2 released to O2 consumed. It is 1 for carbs, and less than 1 for fats and proteins.
Common Student Misconceptions
Misconception 1: “Plants only do photosynthesis during the day and respiration only at night.”
Correction: This is a very common mistake! Plants respire 24 hours a day, 7 days a week. If a living cell stops respiring, it dies. During the day, they are doing both simultaneously. It just seems like they only respire at night because, during the day, the massive oxygen output from photosynthesis completely overshadows the small amount of oxygen they consume for respiration.
Misconception 2: Breathing and Cellular Respiration are the exact same thing.
Correction: Breathing (ventilation) is just the mechanical physical process of moving air in and out of the body to get oxygen to the blood. Cellular Respiration is a microscopic, biochemical series of chemical reactions happening inside every single cell to extract energy from food.
Practice Set: Test Your Knowledge (CBSE Pattern)
Very Short Answer Questions (1 Mark)
Q1. Name the ultimate electron acceptor in aerobic respiration. What does it form after accepting the electrons?
Answer: Molecular oxygen (O2) is the final electron acceptor. After accepting electrons and protons, it gets reduced to form metabolic water (H2O).
Q2. Where exactly does the electron transport system (ETS) operate in a eukaryotic cell?
Answer: The ETS operates on the inner mitochondrial membrane.
Q3. What is the value of the Respiratory Quotient (RQ) when carbohydrates are used as the primary respiratory substrate?
Answer: The RQ is 1.0, because the volume of carbon dioxide released is exactly equal to the volume of oxygen consumed.
Short Answer Questions (2-3 Marks)
Q4. Why is less energy produced during anaerobic respiration (fermentation) compared to aerobic respiration?
Answer: Anaerobic respiration results in the incomplete breakdown of the glucose molecule. The carbon-carbon bonds are not fully oxidized, leaving a lot of potential energy trapped inside the waste products (like lactic acid or ethanol). Additionally, the electron transport chain cannot function without oxygen, so the cell cannot cash in its NADH molecules for the large ATP payoff. Hence, the net yield is only 2 ATP, compared to the theoretical 38 ATP of aerobic respiration.
Q5. Differentiate between Glycolysis and the Krebs Cycle.
Answer:
1. Location: Glycolysis takes place in the cytoplasm, whereas the Krebs cycle takes place in the mitochondrial matrix.
2. Oxygen Requirement: Glycolysis is anaerobic and functions with or without oxygen. The Krebs cycle strictly requires aerobic conditions to proceed.
3. Process: Glycolysis is a linear pathway that breaks down glucose into pyruvic acid. The Krebs cycle is a cyclic pathway that completely oxidizes Acetyl CoA into carbon dioxide.
Q6. State the role of ATP synthase (Complex V) in mitochondria.
Answer: ATP synthase is a membrane-bound enzyme complex consisting of F0 and F1 components. It acts like a channel. The ETS pumps protons into the inter-membrane space, creating a high-energy concentration gradient. As these protons forcefully flow back down their gradient through the F0 channel of ATP synthase, the energy is used by the F1 headpiece to catalyze the bonding of ADP and inorganic phosphate, synthesizing ATP. This process is called chemiosmosis.
Long Answer Questions (5 Marks)
Q7. Explain why the respiratory pathway is better described as an amphibolic pathway rather than simply a catabolic one.
Answer: Traditionally, respiration was defined purely as a catabolic pathway because its primary function is the breakdown of complex fuel molecules (like glucose, fats, or proteins) into simpler substances (CO2 and H2O) to release energy.
However, this is only half the story. The living cell is highly efficient and uses the intermediate compounds generated during this breakdown process as raw building blocks to synthesize new complex molecules when needed (anabolism).
For example:
– If the body needs to store energy as fat, it will stop the oxidation of Acetyl CoA and withdraw it from the pathway to synthesize fatty acids.
– If the body needs to make certain amino acids for protein synthesis, it will withdraw compounds like alpha-ketoglutaric acid from the Krebs cycle.
Because the pathway acts as a central hub that facilitates BOTH the breakdown of molecules for energy (catabolism) and the synthesis of molecules for growth (anabolism), it is scientifically accurate to call it an ‘amphibolic’ pathway rather than strictly catabolic.
Q8. Trace the complete journey of a glucose molecule through aerobic respiration, highlighting the locations and the energy carriers produced at each major step.
Answer: The complete oxidation of glucose happens in several distinct stages:
1. Glycolysis (Cytoplasm): The 6-carbon glucose is broken down through 10 enzymatic steps into two molecules of 3-carbon Pyruvic Acid. This yields a net of 2 ATP and 2 NADH molecules.
2. Link Reaction (Mitochondrial Matrix): The pyruvic acid enters the mitochondria and undergoes oxidative decarboxylation. It loses CO2 and forms the 2-carbon Acetyl CoA. Since there are two pyruvates, this step yields 2 NADH molecules.
3. Krebs Cycle (Mitochondrial Matrix): Acetyl CoA enters the cyclic pathway by joining OAA. Through a series of decarboxylations and oxidations, the carbon structure is completely dismantled into CO2. For one original glucose molecule (two turns of the cycle), this yields 6 NADH, 2 FADH2, and 2 ATP (via GTP).
4. Electron Transport System (Inner Mitochondrial Membrane): All the accumulated high-energy carriers (10 NADH and 2 FADH2) deliver their electrons to the ETS. The electrons cascade down the protein complexes, pumping protons to create a gradient. ATP synthase uses this gradient to generate a massive payoff of ATP. Oxygen accepts the tired electrons at the end, forming water.
This entire coordinated journey results in the theoretical maximum net gain of 38 ATP molecules.
Case-Based / Competency-Based Question (4 Marks)
Q9. Read the situation and answer the questions.
A group of researchers isolates mitochondria from a plant cell and places them in a heavily oxygenated buffer solution. They supply the mitochondria with a continuous feed of Pyruvic acid. They notice that the Krebs cycle runs efficiently and a large amount of ATP is produced. Suddenly, the researchers add a chemical toxin to the solution that tightly binds to and blocks oxygen, preventing it from interacting with any enzymes.
(a) Which specific component of the mitochondrial machinery will shut down first due to the lack of available oxygen?
(b) Explain why the Krebs cycle, which doesn’t directly use oxygen in its reactions, will also rapidly grind to a halt in this situation.
(c) If the intact plant cell was placed in this toxic environment, what alternative pathway would it have to rely on for survival, and what would the end product be?
Answer:
(a) The terminal complex of the Electron Transport System (Complex IV) will shut down first because oxygen acts as the final electron acceptor.
(b) Even though the Krebs cycle doesn’t use O2 directly, it requires a constant supply of empty NAD+ and FAD+ carriers to accept hydrogen. Since the ETS is blocked (due to no oxygen), the loaded NADH and FADH2 cannot unload their electrons. All carriers remain full, leaving the Krebs cycle with no empty carriers to proceed, causing it to stop.
(c) The intact cell would be forced to revert to anaerobic respiration (Fermentation) in the cytoplasm to survive. In a plant cell, this would be alcoholic fermentation, resulting in the production of ethanol and carbon dioxide, yielding very little ATP.
Assertion-Reason Question
Q10. For the following question, two statements are given—one labeled Assertion (A) and the other labeled Reason (R). Select the correct answer from the codes (a), (b), (c), and (d) as given below.
(a) Both A and R are true, and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true, but R is false.
(d) A is false, but R is true.
Assertion (A): ATP acts as the universal energy currency of the cell.
Reason (R): The energy released from the oxidation of food is not used directly by the cell, but is trapped in the chemical bonds of ATP to be utilized whenever and wherever needed.
Answer: (a). The assertion is completely correct, and the reason perfectly explains why ATP is referred to as currency—it acts as a standardized, easily transportable storage unit for energy derived from food breakdown, ready to be “spent” on cellular work.
End of Notes.
Students, remember that understanding the logical flow of these energy pathways is much easier than memorizing the chemical structures. Focus on the inputs, the locations, and the final products! Happy studying!
Read Also:
Chapter 11- Photosynthesis in Higher Plants
For official syllabus and textbooks, visit the
Official Educational Boards Website.