Chapter 4: Carbon and its Compounds
Start Chapter MCQ QuizSimplified Notes: Carbon and its Compounds
Class 10 Science | Chapter 4 Carbon and its Compounds class 10 NCERT solutions| In-Depth Analysis
1. Meet Carbon: The Building Block of Life
Dear Student Carbon is a very special and important element in our world, and even though it is not found in large amounts in the Earth’s crust or in the air, it plays a huge role in our daily life.
You can think of carbon as the backbone of life because all living things, such as plants, animals, and human beings, are made up of carbon compounds.
Our body tissues, the food we eat, and even the air we breathe during respiration are connected to carbon in some way. Not only living things, but many non-living things we use every day also depend on carbon. For example, the clothes we wear, the fuels we use for cooking and transportation, the plastic items in our homes, and the medicines we take are all made from carbon-based substances.
This shows how deeply carbon is linked to our daily activities. One of the main reasons carbon is so important is its ability to form bonds with other elements and also with itself, which helps it create millions of different compounds. Because of this property, carbon is called a versatile element. In this chapter, we will study these special features of carbon in detail and understand how one element can form so many useful substances that make our life comfortable and convenient.
2. The Secret of Carbon’s Friendliness: Covalent Bonding
Unlike metals that give away electrons, carbon prefers to share them. This sharing of electrons between atoms creates a strong connection called a covalent bond.
- Why Carbon Shares: Carbon has four electrons in its outer shell. Losing or gaining four electrons is difficult, so the perfect solution is to share its four electrons with other atoms, forming four stable covalent bonds.
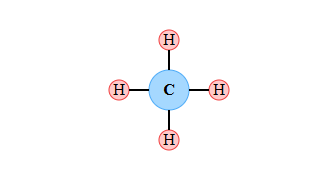
Covalent Bonding in Methane (CH4) C H H H H
- Single, Double, and Triple Handshakes:
- Single Bond: One pair of shared electrons (e.g., H-H).
- Double Bond: Two pairs of shared electrons (e.g., O=O).
- Triple Bond: Three pairs of shared electrons (e.g., N≡N).
- Properties of Covalent Compounds: Covalent compounds do not conduct electricity like plastic. They melt easily like butter and also boil at low temperature, just like water boiling faster in open pot.
3. Carbon’s Two Amazing Superpowers
Two key properties allow carbon to form millions of different compounds.
- Catenation: The Power of Self-Linking
Catenation means carbon atoms can easily join with each other. Like children holding hands to make a long line or circle, carbon forms long chains and rings. Other elements cannot do this so well. - Tetravalency: The Power of Four Connections
Tetravalency means carbon has four outer electrons, like four hands. Using these electrons, one carbon atom can join with four other atoms. This helps carbon make many different and strong molecules.
Carbon’s Different Costumes: Allotropes

Carbon can exist in different physical forms, called allotropes, where the atoms are arranged differently. Allotropes of Carbon: Diamond and Graphite Diamond (Rigid 3D) Graphite (Flat Layers)
- Diamond: Each carbon atom is bonded to four others in a rigid 3D structure, making it the hardest natural substance.
- Graphite: Each carbon atom is bonded to three others in flat hexagonal layers that can slide over each other. This makes graphite soft and slippery and a good conductor of electricity.
- Fullerenes: The most famous is Buckminsterfullerene (C-60), which looks like a soccer ball.
4. The Hydrocarbon Family Tree
Compounds containing only carbon and hydrogen are called hydrocarbons.
- Saturated Hydrocarbons (Alkanes): Contain only single bonds. They are generally less reactive. The general formula is
CₙH₂ₙ₊₂. (e.g., Methane, Ethane). - Unsaturated Hydrocarbons: Contain at least one double or triple bond and are more reactive.
- Alkenes: Contain one or more double bonds. General formula:
CₙH₂ₙ. (e.g., Ethene). - Alkynes: Contain one or more triple bonds. General formula:
CₙH₂ₙ₋₂. (e.g., Ethyne).
- Alkenes: Contain one or more double bonds. General formula:
Structural Isomers: Same Formula, Different Shapes
These are compounds that have the identical molecular formula but different structural arrangements of atoms. For example, butane (C₄H₁₀) can exist as a straight chain or a branched chain.
5. Adding Personality: Functional Groups & Naming
The properties of a carbon compound can be changed by replacing a hydrogen atom with a heteroatom or a group of atoms. This group is called a functional group and it dictates the molecule’s chemical personality.
| Functional Group | Formula | Suffix/Prefix | Example |
|---|---|---|---|
| Alcohol | -OH | -ol | Propanol |
| Aldehyde | -CHO | -al | Hexanal |
| Ketone | -C=O | -one | Butanone |
| Carboxylic Acid | -COOH | -oic acid | Ethanoic acid |
| Halogen | -Cl, -Br | Chloro-, Bromo- | Chloropropane |
The Homologous Series: A Family with a Pattern
This is a series of compounds with the same functional group where each successive member differs by a -CH₂ unit. Members have similar chemical properties, but their physical properties (like boiling point) show a gradual increase with mass.
6. Carbon in Action: Key Chemical Reaction
Saturated hydrocarbons tend to burn with a clean flame, while unsaturated ones often produce a sooty flame.
- Combustion: Carbon compounds burn in oxygen to release energy, CO₂, and water.
- Oxidation: Alcohols can be converted to carboxylic acids using oxidizing agents like alkaline KMnO4.
- Addition Reaction: Characteristic of unsaturated compounds. Hydrogen is added across the double/triple bond using a catalyst (hydrogenation), for example, to convert vegetable oils into solid fats.
- Substitution Reaction: Characteristic of saturated compounds. One or more hydrogen atoms are replaced by another atom, like chlorine, in the presence of sunlight.
7. Two Famous Carbon Compounds
- Ethanol (C2H5OH): The active ingredient in alcoholic beverages and a good solvent.
- Ethanoic Acid (CH3COOH): Commonly known as acetic acid; a dilute solution is called vinegar. It reacts with an alcohol to form a sweet-smelling ester (esterification reaction).
8. The Science of Cleaning: Soaps & Detergents
Soap molecules have two ends: a hydrophilic (water-loving) head and a hydrophobic (water-fearing) tail. In water, they form clusters called micelles, with the tails pointing inwards to trap oily dirt and the heads pointing outwards.
Soap molecules form micelles to trap and wash away oily dirt.
- Hard Water Woes: In hard water, soap reacts with calcium and magnesium salts to form insoluble sticky scum.
- Why Detergents Win: Detergents do not form scum with the ions in hard water, so they remain effective cleaning agents.
9. Practice Questions (Original)
Multiple Choice Questions (MCQ)
- A student notices that a cooking oil solidifies when hydrogen gas is passed through it. What does this demonstrate?
(a) Oxidation (b) Substitution (c) An addition reaction (d) Esterification
Answer: (c) An addition reaction - Which pair represents a homologous series?
(a) CH4 and C2H4 (b) C2H5OH and C3H7OH (c) C3H4 and C3H8 (d) CH3COOH and CH3CHO
Answer: (b) C2H5OH and C3H7OH - Graphite is used as a lubricant and an electrode. Which two properties make this possible?
(a) Hardness and insulation (b) Slipperiness and conductivity (c) Lustre and density (d) Brittleness and inertness
Answer: (b) Slipperiness and electrical conductivity - A compound has the formula C4H8. It reacts with hydrogen to form C4H10. The original compound is an:
(a) Alkane (b) Alkyne (c) Alcohol (d) Alkene
Answer: (d) Alkene - Why does soap form a cloudy solution in water?
(a) It is insoluble. (b) It forms micelles large enough to scatter light. (c) It reacts with water. (d) It is a colored compound.
Answer: (b) It forms micelles that are large enough to scatter light.
Short Answer Questions
- Q6. Diamond is extremely hard while graphite is soft. Briefly explain why.
A6. In diamond, each carbon atom is bonded to four others in a rigid 3D network. In graphite, carbon atoms are bonded in flat layers that can slide past one another. - Q7. What are structural isomers? Draw the two isomers of butane (C4H10).
A7. Isomers have the same molecular formula but different structures. The two isomers are n-butane (straight-chain) and isobutane (branched-chain). - Q8. Why is a mixture of ethyne and pure oxygen used for welding instead of ethyne and air?
A8. Burning ethyne in pure oxygen results in complete combustion, producing a very hot, clean flame suitable for welding. Using air would lead to incomplete combustion and a cooler, sooty flame.
Long Answer Questions
- Q11. You are given ethanol, ethanoic acid, and a soap solution. Describe simple tests to identify each.
A11. 1. Use blue litmus paper: only ethanoic acid will turn it red. 2. Add sodium bicarbonate: only ethanoic acid will produce fizzing (CO₂). 3. Shake with water: soap solution will form a lather, while ethanol will form a clear solution. - Q12. Explain the concept of a homologous series using alcohols as an example. List three characteristics.
A12. A homologous series is a family of compounds with the same functional group where members differ by a -CH2 unit. Example: Methanol (CH3OH), Ethanol (C2H5OH), Propanol (C3H7OH). Characteristics: 1. Represented by a general formula (CnH2n+1OH). 2. Successive members differ by a -CH2 group. 3. Similar chemical properties but gradually changing physical properties. - Q13. What is catenation? Explain why carbon exhibits this property to a large extent, while silicon does not.
A13. Catenation is the self-linking ability of an element. Carbon excels at this due to the high strength of the C-C bond and its small atomic size. Silicon atoms are larger, making the Si-Si bond weaker and less stable, so it can only form short, reactive chains.
Read Also: Class 10 Chapter 3- Metals and Non-metals
For more check official website of NCERT