Chapter 3: Metals and Non-metals
1. Introduction: Meeting the Elements | Metals and non-metals NCERT solutions
Hello students! Today we are going to explore the building blocks of everything around us—Elements. Imagine you have a giant box of Lego bricks. In chemistry, there are currently 118 different types of these bricks known to us. We call them elements.
Now, if you look at these 118 elements, they aren’t all the same. Some are shiny and hard, like the gold in a ring or the iron in a gate. Others are invisible gases like the oxygen we breathe or dull powders like coal (carbon). To make studying them easier, scientists have divided them into two main “teams” or families:
- Metals: The shiny, hard, and conductive group (like Iron, Copper, Gold).
- Non-metals: The dull, brittle, or gaseous group (like Oxygen, Carbon, Sulfur).
There is also a tiny third group called Metalloids that act a bit like both, but in this chapter, our main heroes are the Metals and Non-metals. Let’s get to know them better!
2. Physical Properties: The “Look and Feel” Test
How do you know if something is a metal or a non-metal without doing any chemistry experiments? You just look at it and touch it! This is called observing physical properties.

Notice how metals shine and can be shaped into different forms without breaking.
A. Properties of Metals
Think of metals as the “tough guys” of the periodic table. Here is what makes them special:
- Metallic Lustre (Shine): Metals love to show off! In their pure state, they have a shining surface. That’s why we use gold and silver for jewelry.
- Hardness: Try pressing a piece of iron with your finger. Nothing happens, right? Metals are generally very hard. But wait—there is a catch! Metals like Sodium and Potassium are so soft you can cut them with a kitchen knife like a block of paneer.
- Malleability (Beating into sheets): If you hit a piece of coal with a hammer, it shatters into powder. But if you hit a piece of iron or gold, it flattens out. This property of turning into thin sheets is called Malleability. Gold and Silver are the champions of this; that’s how we get those super thin silver foils on sweets (Kaju Katli)!
- Ductility (Drawing into wires): Have you ever seen a wooden wire? No, right? That’s because only metals can be stretched into thin wires. This is called Ductility. Gold is surprisingly the most ductile metal—from just 1 gram of gold, you can make a wire that is 2 kilometers long!
- Conductivity (Heat & Electricity): Metals are like highways for heat and electricity. This is why cooking pans are made of metal (to carry heat to food) and electrical wires are made of copper (to carry electricity). Lead and Mercury are the lazy ones here; they are poor conductors compared to others.
- Sonority (The Sound): When you drop a metal spoon, it goes “TANG!”. This ringing sound is called sonorous. That is exactly why school bells and temple bells are made of metal.
B. Properties of Non-Metals
Non-metals are the complete opposite. They are usually either gases or solids that break easily.
- They don’t shine (Dull).
- They break if you hit them (Brittle).
- They don’t let electricity or heat pass through (Insulators).
- They don’t make a ringing sound (Non-sonorous).
C. The “Rule Breakers” (Exceptions)
Chemistry is full of exceptions. You must remember these for your exams because examiners love asking about them!
| Property | General Rule | The Exception (Memorize This!) |
|---|---|---|
| State of Matter | Metals are solid. | Mercury is a liquid metal at room temperature. |
| Lustre (Shine) | Non-metals are dull. | Iodine is a non-metal but it is shiny (lustrous). |
| Hardness | Non-metals are soft/brittle. | Diamond (a form of Carbon) is the hardest natural substance known! |
| Conductivity | Non-metals are insulators. | Graphite (another form of Carbon) conducts electricity very well. |
| Melting Point | Metals have high melting points. | Gallium and Cesium have such low melting points they will melt on your palm! |
3. Chemical Properties: How They React
Now let’s get into the lab (mentally!). We are going to see how metals behave when they meet Oxygen, Water, and Acids.
A. What happens when Metals are burnt in Air?
Almost all metals combine with oxygen to form Metal Oxides.
Metal + Oxygen → Metal Oxide
For example, when you heat Copper wire, it doesn’t burn, but it gets coated with a black layer. That black stuff is Copper Oxide.
Equation: 2Cu + O₂ → 2CuO (Black color)
Are all oxides the same?
Usually, metal oxides are basic. They turn red litmus to blue. But some metal oxides are “two-faced.” They react with both acids AND bases to produce salt and water. We call these Amphoteric Oxides. Zinc Oxide and Aluminum Oxide are the best examples.
Example: Aluminum Oxide reacting with an Acid (acting like a base):
Al₂O₃ + 6HCl → 2AlCl₃ + 3H₂O
Example: Aluminum Oxide reacting with a Base (acting like an acid):
Al₂O₃ + 2NaOH → 2NaAlO₂ (Sodium Aluminate) + H₂O
B. What happens when Metals meet Water?
This is interesting because different metals treat water differently.
- The Crazy Reactors: Sodium and Potassium react violently with cold water. They produce hydrogen gas which immediately catches fire. It’s dangerous!
- The Floater: Calcium reacts less violently. The hydrogen bubbles formed stick to the surface of the metal, making the Calcium float on water.
- The Hot Water Fans: Magnesium doesn’t react with cold water. It needs hot water to react. It also floats due to bubbles.
- The Steam Lovers: Aluminum, Iron, and Zinc are too lazy to react with liquid water (hot or cold). They only react with steam (gas).
- The Non-Reactors: Lead, Copper, Silver, and Gold do not react with water at all. That’s why water pipes are often made of copper or plastic, not iron (which rusts).
C. What happens when Metals meet Acids?
Generally, a Metal + Dilute Acid → Salt + Hydrogen Gas.
If you bring a burning matchstick near the gas produced, it burns with a “pop” sound. That is the test for Hydrogen gas.
Note: Hydrogen gas is NOT evolved when a metal reacts with Nitric Acid (HNO₃). Why? Because Nitric Acid is a strong oxidizing agent. It turns the hydrogen produced into water immediately!
D. The Displacement Reaction
This is like a wrestling match. A stronger (more reactive) metal will kick out a weaker (less reactive) metal from its solution.
If you put an Iron nail into a blue Copper Sulphate solution, the blue color fades to green. Why? Because Iron is stronger than Copper. It displaces Copper and takes its place.
Equation: Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s)
4. The Reactivity Series (The League Table)
Scientists arranged metals in a list from the “Most Reactive” at the top to the “Least Reactive” at the bottom. This list is super useful to predict if a reaction will happen or not.
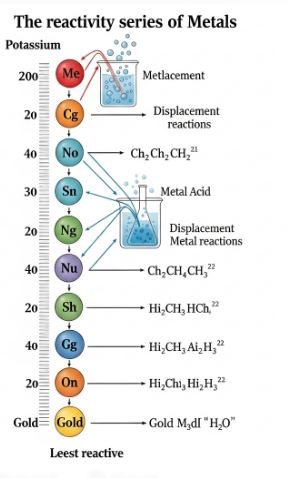
Potassium is the King (Top), and Gold is at the bottom.
How to memorize this? Here is a funny mnemonic for you:
“Please Stop Calling Me A Zebra, I Like Her Calling Me Smart Guy.”
- Potassium (Most Reactive)
- Sodium
- Calcium
- Magnesium
- Aluminum
- Zinc
- Iron
- Lead
- Hydrogen (Non-metal, but used for comparison)
- Copper
- Mercury
- Silver
- Gold (Least Reactive)
5. How do Metals and Non-Metals Bond?
Why do elements react? Because they want to be stable. In chemistry, being “stable” usually means having 8 electrons in the outermost shell (like the Noble Gases).
The Give and Take Relationship
- Metals have extra electrons (1, 2, or 3) in their outer shell. They want to lose them. When they lose negative electrons, they become Positive Ions (Cations).
- Non-metals are missing electrons (5, 6, or 7). They want to gain electrons to reach 8. When they gain negative electrons, they become Negative Ions (Anions).
Example: Sodium Chloride (Common Salt)
Sodium (Na) has 1 extra electron. Chlorine (Cl) needs 1 electron. Sodium gives its electron to Chlorine. Now Sodium becomes positive (Na+) and Chlorine becomes negative (Cl-). Opposites attract! They stick together strongly. This bond is called an Ionic Bond.
Properties of Ionic Compounds
- Physical Nature: They are solid and hard because of the strong force of attraction between positive and negative ions.
- Melting Point: You need a lot of heat to break that strong bond, so they have very high melting and boiling points.
- Solubility: They usually dissolve in water but not in oils or kerosene.
- Conduction of Electricity: This is crucial! In solid form, ions are locked in place, so they don’t conduct electricity. But if you melt them or dissolve them in water, the ions become free to move, and they conduct electricity.
6. Metallurgy: Extracting Metals from Earth
You don’t just find an Iron rod lying in the ground. Metals are usually found mixed with sand, rocks, and other chemicals. These mixtures are called Minerals. If a mineral has enough metal that can be extracted profitably, we call it an Ore.
The process of getting pure metal from ore is called Metallurgy. It has three main steps:
Step 1: Enrichment of Ore
First, we remove the “Gangue”. Gangue is just a fancy word for earthly impurities like sand, soil, and clay found in the ore. We wash it or use chemicals to clean it.
Step 2: Extracting the Metal (Based on Reactivity)
A. Extracting Metals low in activity series (like Mercury, Copper)
These are easy. Often, just heating them in air is enough.
Example: Cinnabar (HgS) is an ore of Mercury. When heated, it turns to oxide, and on further heating, it turns to pure Mercury.
B. Extracting Metals in the middle (Zinc, Iron, Lead)
These are usually found as Carbonates or Sulfides. It is easier to get metal from its Oxide, so we first convert them to oxides.
| Roasting | Calcination |
|---|---|
| Used for Sulfide ores. | Used for Carbonate ores. |
| Ore is heated in excess air. | Ore is heated in limited air. |
| Equation: 2ZnS + 3O₂ → 2ZnO + 2SO₂ | Equation: ZnCO₃ → ZnO + CO₂ |
Once we have the Oxide (ZnO), we use Carbon (Coke) to remove the oxygen. This is called Reduction.
ZnO + C → Zn + CO
C. Extracting Metals at the top (Sodium, Aluminum)
These metals love oxygen so much that Carbon cannot pull the oxygen away from them. So, we use electricity. This method is called Electrolytic Reduction.
Step 3: Refining (Purification)
The metal we get is still not 100% pure. We use electricity to clean it up. This is Electrolytic Refining.
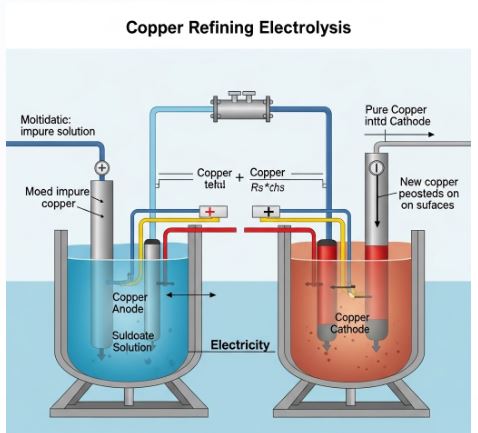
The Setup:
- Anode (+): A thick block of IMPURE metal.
- Cathode (-): A thin strip of PURE metal.
- Electrolyte: Salt solution of the metal (e.g., Copper Sulphate).
The Magic: When you switch on the current, pure metal from the Anode dissolves into the water and then goes to stick onto the Cathode. All the dirt falls to the bottom of the Anode. We call this dirt “Anode Mud”.
7. Corrosion: The Slow Death of Metals
Have you seen reddish-brown powder on old iron gates? That is Rust. Corrosion is a natural process where metals get “eaten up” by air, moisture, or acids.
- Silver turns black because it reacts with Sulfur in the air to form Silver Sulfide.
- Copper turns green because it reacts with moist Carbon Dioxide to form Copper Carbonate.
- Iron turns reddish-brown because it forms hydrated Iron Oxide (Rust).
Prevention: How to stop it?
- Painting/Oiling: Creates a barrier so air cannot touch the metal.
- Galvanization: We put a thin layer of Zinc over Iron. Zinc is the bodyguard; it sacrifices itself to protect Iron.
- Alloying: Mixing metals improves their quality.
8. Alloys: Better Together
An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal. It changes the properties of the metal.
- Steel: Iron is too soft. Mix it with a little Carbon (0.05%), and it becomes hard and strong steel.
- Stainless Steel: Mix Iron with Nickel and Chromium. Result? No rusting!
- Brass: Copper + Zinc.
- Bronze: Copper + Tin.
- Solder: Lead + Tin. It has a low melting point, perfect for joining electrical wires.
- Amalgam: Any alloy where one metal is Mercury.
Gold Purity: Pure Gold is 24 Carat. It is very soft. Jewelry is usually made of 22 Carat Gold (22 parts gold + 2 parts silver/copper) to make it hard enough to hold its shape.
9. Comprehensive Question Bank
If you can answer these, you are ready for the exam!
Part A: Multiple Choice Questions (MCQs)
- Which of the following pairs will give displacement reaction?
(a) NaCl solution and copper metal
(b) MgCl₂ solution and aluminum metal
(c) FeSO₄ solution and silver metal
(d) AgNO₃ solution and copper metal
Answer: (d) Copper is more reactive than Silver, so it will displace Silver. In all other options, the metal added is less reactive than the metal in the solution. - Which of the following methods is suitable for preventing an iron frying pan from rusting?
(a) Applying grease (b) Applying paint (c) Applying a coating of zinc (d) All of the above
Answer: (c) We can’t use grease or paint on a frying pan because heat will burn them. Zinc coating (Galvanization) is the best option. - An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be:
(a) Calcium (b) Carbon (c) Silicon (d) Iron
Answer: (a) Calcium reacts with oxygen to form Calcium Oxide. It is ionic (high melting point) and dissolves in water to form limewater. Carbon forms a gas. Silicon and Iron oxides are insoluble. - Food cans are coated with tin and not with zinc because:
(a) Zinc is costlier than tin (b) Zinc has a higher melting point (c) Zinc is more reactive than tin (d) Zinc is less reactive than tin
Answer: (c) Zinc is more reactive. It might react with the organic acids in the food and spoil it. Tin is less reactive and safe. - Which metal is found in liquid state at room temperature?
(a) Iron (b) Mercury (c) Gold (d) Aluminum
Answer: (b) Mercury. - The most ductile metal is:
(a) Silver (b) Gold (c) Copper (d) Aluminum
Answer: (b) Gold. You can draw a wire of 2km from just 1g of gold. - Reaction between X and Y forms compound Z. X loses electron and Y gains electron. Which of the following properties is NOT shown by Z?
(a) High melting point (b) Low melting point (c) Conducts electricity in molten state (d) Occurs as solid
Answer: (b) Since electrons are transferred, Z is an ionic compound. Ionic compounds have HIGH melting points, not low.
Part B: Short Answer Questions (2-3 Marks)
Q1. Give reasons: Platinum, Gold, and Silver are used to make jewelry.
Answer: There are two main reasons:
1. They are highly malleable and ductile, so they can be easily shaped into intricate designs.
2. They are the least reactive metals. They do not corrode or tarnish easily, so they keep their shine for a very long time.
Q2. Why is Sodium kept immersed in kerosene oil?
Answer: Sodium is a very “angry” metal (highly reactive). If kept in the open, it reacts with oxygen and catches fire. It also reacts violently with moisture in the air. Kerosene oil cuts off its contact with air and moisture, keeping it safe.
Q3. Differentiate between Mineral and Ore.
Answer:
– Mineral: Any natural material found in the earth’s crust containing metals is a mineral.
– Ore: Those specific minerals from which metal can be extracted profitably and conveniently are called ores. All ores are minerals, but not all minerals are ores.
Q4. Explain the formation of Magnesium Chloride (MgCl₂) with the help of electron dot structure.
Answer:
– Magnesium (Atomic No. 12) has configuration 2, 8, 2. It wants to lose 2 electrons.
– Chlorine (Atomic No. 17) has configuration 2, 8, 7. It wants to gain 1 electron.
– Since Mg needs to lose 2, but one Cl can only take 1, Magnesium gives one electron to one Chlorine atom and the second electron to another Chlorine atom.
– Result: Mg²⁺ and two Cl⁻ ions form MgCl₂.
Q5. What are amphoteric oxides? Give two examples.
Answer: Oxides that show acidic as well as basic behavior are called amphoteric oxides. This means they react with both acids and bases to produce salt and water.
Examples: Aluminum Oxide (Al₂O₃) and Zinc Oxide (ZnO).
Part C: Long Answer Questions (5 Marks)
Q1. Explain the process of electrolytic refining of copper with a diagram.
Answer: Electrolytic refining is used to get very pure copper.
Apparatus: A tank containing acidified copper sulphate solution (electrolyte).
Electrodes:
1. Anode: A thick block of impure copper.
2. Cathode: A thin strip of pure copper.
Process: When electric current is passed:
– Copper atoms from the impure anode lose electrons and enter the solution as Copper ions (Cu²⁺).
– These Cu²⁺ ions move towards the cathode (negative electrode).
– At the cathode, they gain electrons and deposit as pure copper metal.
– Impurities like gold and silver settle down at the bottom of the anode as ‘Anode Mud’.
Reaction:
Anode: Cu → Cu²⁺ + 2e⁻
Cathode: Cu²⁺ + 2e⁻ → Cu
Q2. A metal ‘E’ is stored under kerosene. When a small piece of it is left open in air, it catches fire. When the product formed is dissolved in water, it turns red litmus blue.
(i) Name the metal ‘E’.
(ii) Write the chemical equation for the reaction with air.
(iii) Write the equation for the reaction with water.
Answer:
(i) The metal is Sodium (Na) (or Potassium). We know this because it is stored in kerosene and catches fire in air.
(ii) Reaction with Oxygen: 4Na(s) + O₂(g) → 2Na₂O(s) (Sodium Oxide).
(iii) Sodium Oxide dissolves in water to form an alkali: Na₂O(s) + H₂O(l) → 2NaOH(aq). The product NaOH is basic, which turns red litmus blue.
Q3. (a) Define corrosion. (b) What is corrosion of iron called? (c) How will you prevent corrosion of iron?
Answer:
(a) Corrosion is the slow eating away of metals by the action of air, moisture, or chemicals on their surface.
(b) Corrosion of iron is specifically called Rusting. The formula of rust is Fe₂O₃.xH₂O.
(c) Methods to prevent rusting:
1. Painting: Apply paint on gates and railings.
2. Greasing/Oiling: Used for tools and machine parts.
3. Galvanization: Coating iron objects with a layer of Zinc. Even if the zinc coating scratches, it still protects the iron because zinc is more reactive.
4. Chrome Plating: Used in bicycle handles and car parts for a shiny look and protection.
5. Alloying: Making Stainless Steel (Iron + Nickel + Chromium).
Part D: Give Reasons (HOTS)
- Why do ionic compounds have high melting points?
Because the force of attraction between the positive and negative ions is very strong. A lot of energy (heat) is needed to break this force. - Why does calcium float in water?
When calcium reacts with water, hydrogen gas bubbles are formed. These bubbles stick to the surface of the calcium metal, acting like a life-jacket, making it float. - Why are electric wires made of copper?
Copper is a very good conductor of electricity (low resistance) and is also ductile (can be drawn into wires). It is also cheaper than Silver (the best conductor).
Read Also:
Class 10 Chapter 2- Acid, Base, and Salts
For more check official website of
NCERT
