1. Introduction: The Dance of the Molecules | Class 11 Physics Chapter 12 Kinetic theory of Gases notes
Hello everyone! Welcome to Chapter 12. Up until now, we have been studying matter from the outside. We’ve looked at blocks sliding down ramps, planets orbiting stars, and fluids flowing through pipes. In all those chapters, we treated objects as solid, continuous chunks of stuff. But today, we are going to put on our “microscopic glasses” and look inside.
What we are going to find is chaos. Beautiful, organized chaos.
The Kinetic Theory of Gases is essentially the story of how the frantic motion of billions of tiny invisible particles (atoms and molecules) creates the calm, steady properties we observe in everyday life—like the pressure in a tire or the temperature of your tea. It is a bridge. It bridges the gap between the Microscopic World (atoms, speeds, collisions) which we cannot see, and the Macroscopic World (Pressure, Volume, Temperature) which we can measure.
1.1 Historical Context
It wasn’t always obvious that matter was made of atoms. For centuries, people thought air was a continuous fluid. Scientists like John Dalton (Atomic Theory), James Clerk Maxwell, and Ludwig Boltzmann fought hard to establish the kinetic theory. They used statistics to predict how huge crowds of molecules would behave, even if they couldn’t track individual ones. This statistical approach is the foundation of modern physics.
2. Molecular Nature of Matter
Before we dive into the math, let’s establish our “Atomic Hypothesis.” The core ideas that govern this theory are simple but profound.
The Core Postulates:
- Matter is not continuous: It is made of discrete particles called atoms or molecules.
- Empty Space: In a gas, the molecules are very far apart. The actual volume of the molecules is tiny compared to the volume of the container. Most of a gas is just empty vacuum! roughly 99.9% of the volume of a gas at room temperature is empty space.
- Constant Motion: These particles are never still. In solids, they vibrate in place. In liquids, they slide past each other. In gases, they fly freely in straight lines until they hit something.
Avogadro’s Hypothesis
Amedeo Avogadro gave us a crucial rule that connects the micro and macro worlds: “Equal volumes of all gases, at the same temperature and pressure, contain the same number of molecules.”
This implies that the identity of the gas doesn’t matter for its volume behavior! One liter of Hydrogen has the same number of molecules as one liter of Oxygen under the same conditions.
The Mole Concept: Since counting atoms is impossible (numbers are too big), we count in packets called “Moles”. One mole is defined as the amount of substance containing as many entities as there are atoms in 0.012 kg of Carbon-12.
Avogadro Number (NA):
NA = 6.022 × 1023
This is the number of particles in exactly one mole of any substance.
3. Behavior of Gases: The Ideal Gas
Real gases are complicated. Oxygen, Nitrogen, and Helium molecules have shapes, they attract each other slightly (Van der Waals forces), and they take up space. To make the math solvable, physicists invented a simplified model called the Ideal Gas.
3.1 What makes a gas “Ideal”?
An ideal gas is a hypothetical gas that follows these rules perfectly:
- Point Masses: The molecules are infinitely small points. They have mass, but zero volume.
- No Forces: The molecules do not attract or repel each other. They ignore each other completely unless they crash. Potential Energy due to interaction is Zero.
- Elastic Collisions: When they hit each other or the walls, no energy is lost. They bounce off perfectly, like super-bouncy rubber balls.
- Random Motion: They obey Newton’s laws of motion but move in random directions with various speeds.
Why?
1. Low Pressure: Molecules are far apart, so their volume becomes negligible.
2. High Temperature: Molecules move so fast that weak attraction forces don’t have time to affect them.
3.2 The Gas Laws (A Quick Recap)
Before the grand equation, scientists found smaller relationships experimentally:
- Boyle’s Law (T is constant): Pressure is inversely proportional to Volume. If you squeeze a balloon (Volume down), the Pressure goes up. (P ∝ 1/V)
- Charles’s Law (P is constant): Volume is directly proportional to Temperature. If you heat a balloon, it expands. (V ∝ T)
- Gay-Lussac’s Law (V is constant): Pressure is directly proportional to Temperature. If you heat a rigid tank, pressure spikes. (P ∝ T)
3.3 The Ideal Gas Law
Combining all the above, we get the single most famous equation in thermal physics:
PV = nRT
Where:
- P = Pressure (Pascal, Pa)
- V = Volume (m3)
- n = Number of moles (mol)
- R = Universal Gas Constant (8.314 J mol-1 K-1). Note that R is same for ALL gases.
- T = Absolute Temperature (Kelvin)
Sometimes, we want to talk about the number of molecules (N) instead of moles (n). We can rewrite the formula using the Boltzmann Constant (kB), which is basically the gas constant “per molecule”.
PV = N kB T
Where kB = R / NA = 1.38 × 10-23 J K-1.
4. Kinetic Theory: The Microscopic Origin of Pressure
Now, let’s answer the question: What IS Pressure?
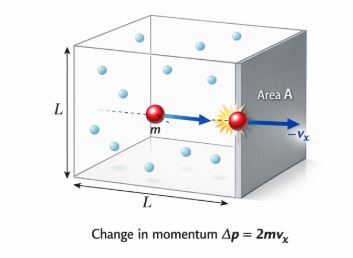
Imagine a cubical container of side L filled with gas. The molecules are flying around randomly. Let’s focus on ONE molecule moving in the X-direction towards the wall.
4.1 The Derivation Logic
- Momentum Change: A molecule with mass m and velocity vx hits the wall. It bounces back with velocity -vx.
Change in momentum (Δp) = Final – Initial = (-mvx) – (mvx) = -2mvx.
Momentum transferred to the wall = +2mvx. - Time Between Collisions: The molecule must travel to the other wall and back to hit the first wall again. Distance = 2L. Time = Distance/Speed = 2L/vx.
- Force: Force is the rate of change of momentum (Newton’s 2nd Law).
Force exerted by ONE molecule = Δp / Δt = (2mvx) / (2L/vx) = mvx2 / L. - Total Force: We sum this force for all N molecules. Since motion is random, the average velocity in X, Y, and Z directions is equal. So vx2 = (1/3)v2.
- Pressure: Pressure = Force / Area. After doing the summation math, we arrive at this beautiful equation:
P = (1/3) n m v²
Wait! Let’s clarify these terms carefully:
– n: Number density (number of molecules per unit volume, N/V).
– m: Mass of one single molecule.
– v²: This is actually the Mean Square Speed (average of the squares of speeds). It is usually written as $\bar{v^2}$.
Alternatively, since total mass M = N × m, and Density ρ = M/V:
P = (1/3) ρ v²
5. Kinetic Interpretation of Temperature
This is arguably the most important concept in the chapter. We know from daily life that “Temperature” measures hotness. But what is it microscopically?
Let’s rearrange our pressure equation from above.
We derived: PV = (1/3) N m v²
We also know the Ideal Gas Law: PV = N kB T
Equating the right sides:
(1/3) N m v² = N kB T
Canceling N and rearranging for Kinetic Energy (1/2 mv²):
Average Kinetic Energy (E) = (3/2) kB T
The Big Revelation
This formula tells us exactly what Temperature is.
Temperature is simply a measure of the average kinetic energy of the molecules.
- If T = 0 (Absolute Zero), the molecules theoretically stop moving (KE = 0).
- Independence of Mass: Notice that ‘m’ (mass) is not in the final equation E = (3/2)kBT. This means at the same temperature (say, 300K), a heavy Oxygen molecule and a light Hydrogen molecule have the exact same average kinetic energy.
Question: Do they have the same speed?
Answer: No! Since KE is same, the lighter molecule (Hydrogen) must move much faster to compensate for its small mass.
5.1 Molecular Speeds: vrms
Since molecules have different speeds, which “speed” do we talk about? We usually use the Root Mean Square (rms) speed. It is the square root of the mean of the squares of the speeds.
vrms = √(3 kB T / m) = √(3 RT / M)
Where M is the Molar Mass.
This confirms that lighter gases move faster. This is why Earth’s atmosphere has lost most of its Hydrogen—it moves so fast it escapes gravity!
6. Maxwell-Boltzmann Distribution of Speeds
Do all molecules move at the same speed? No. It’s like a crowded city street. Some people are walking slowly, some are sprinting, but most are walking at a moderate pace. The Maxwell-Boltzmann Distribution curve shows us this variety.
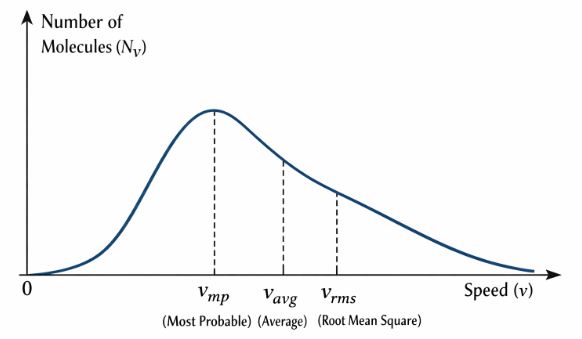
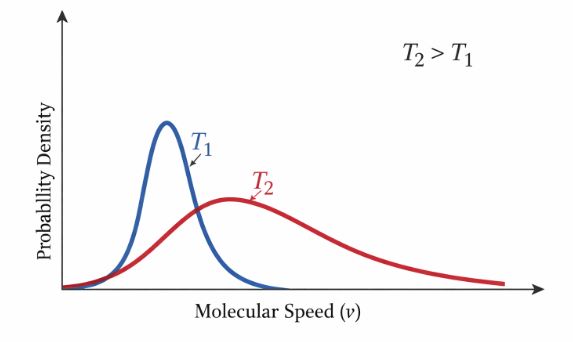
Key Features of the Curve:
1. Bell Shape (Asymmetric): It starts at zero (no molecules at rest), rises to a peak, and has a long tail towards high speeds.
2. Effect of Temperature: As you heat the gas (Temperature rises), the curve flattens and shifts to the right. This means more molecules are moving at higher speeds, and the range of speeds becomes wider.
3. Three Types of Speeds:
– Most Probable Speed (vmp): The speed possessed by the maximum number of molecules (The peak). Formula: √(2RT/M).
– Average Speed (vav): The mathematical average. Formula: √(8RT/πM).
– RMS Speed (vrms): The square root of the mean squared. Formula: √(3RT/M).
Order: vrms > vav > vmp.
7. Degrees of Freedom (f)
So far, we assumed molecules are just dots moving in space. But real molecules can rotate and vibrate too. A Degree of Freedom is an independent way in which a molecule can possess energy. Think of it as a “bank account” where the molecule can store energy.
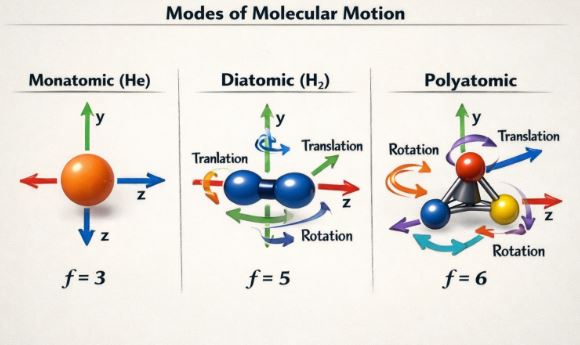
7.1 Types of Motion
- Translational: Moving along X, Y, or Z axes. (Flying through space).
- Rotational: Spinning around an axis.
- Vibrational: Atoms stretching and compressing like springs (only happens at high temperatures).
7.2 Calculating ‘f’
- Monatomic (He, Ar): Can only move in X, Y, Z. It cannot spin effectively (it’s a point) or vibrate.
f = 3 (3 Translational) - Diatomic (H2, O2, N2): Can move in X, Y, Z (3). Can also rotate around two axes perpendicular to the bond (2). Rotation along the bond axis doesn’t count as the moment of inertia is negligible.
f = 5 (3 Trans + 2 Rot) at room temp.
Note: At very high temperatures, it vibrates too, adding 2 more degrees (Kinetic + Potential of vibration), making f = 7. - Polyatomic (H2O, CH4): Can move in X, Y, Z (3). Can rotate around all 3 axes (3).
f = 6 (3 Trans + 3 Rot) + Vibrational modes.
8. Law of Equipartition of Energy
This is a democratic law of physics. It states that nature treats all degrees of freedom equally.
Statement: For any system in thermal equilibrium, the total energy is distributed equally amongst all its degrees of freedom. The energy associated with each degree of freedom per molecule is:
Energy per DoF = (1/2) kB T
This allows us to calculate the Total Internal Energy (U) of one mole of gas easily:
If a gas has f degrees of freedom, energy per molecule is (f/2) kB T.
For one mole (NA molecules), U = NA × (f/2) kB T.
Since NAkB = R, we get:
U = (f/2) R T
9. Specific Heat Capacities
Now we can connect all this theory to something practical: Specific Heat. Remember from Thermodynamics that specific heat is how much energy is needed to raise temperature.
Using the internal energy formula U = (f/2)RT, we can derive the specific heats theoretically.
Molar Specific Heat at Constant Volume (Cv):
This is just the rate of change of U with T.
Cv = dU/dT = (f/2) R
Molar Specific Heat at Constant Pressure (Cp):
From Mayer’s Relation: Cp = Cv + R
Cp = (f/2)R + R = (1 + f/2)R
Ratio of Specific Heats (γ):
γ = Cp / Cv = 1 + (2/f)
Summary Table of Gases
| Gas Type | DoF (f) | Cv | Cp | Gamma (γ) |
|---|---|---|---|---|
| Monatomic | 3 | 1.5 R | 2.5 R | 1.67 |
| Diatomic (Rigid) | 5 | 2.5 R | 3.5 R | 1.40 |
| Diatomic (Vibrating) | 7 | 3.5 R | 4.5 R | 1.28 |
| Polyatomic | 6 | 3.0 R | 4.0 R | 1.33 |
9.1 Specific Heat of Solids
Solids are different. Atoms in a solid cannot translate or rotate; they can only vibrate. A vibration has 2 forms of energy (Kinetic + Potential). In 3D space, an atom has 3 directions of vibration.
So, Degrees of Freedom per atom = 3 × 2 = 6.
According to Equipartition Law:
U = (6/2)RT = 3RT
C = dU/dT = 3R
This matches the Dulong-Petit Law, which states that molar heat capacity of solids is approx 3R (~25 J/mol K) at high temperatures.
10. Mean Free Path
If gas molecules move at speeds of hundreds of meters per second (faster than a race car), why does the smell of perfume take several seconds to cross a room? Why isn’t it instant?
The answer is Collisions.
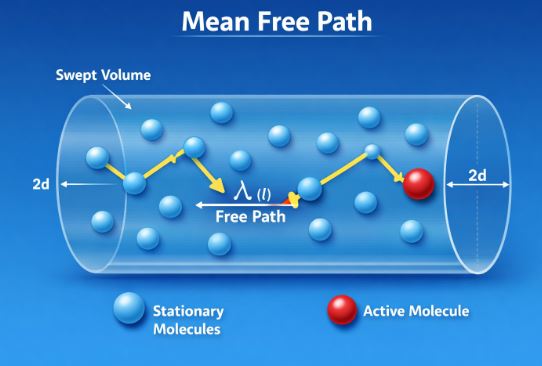
A gas molecule cannot fly straight for very long. It hits another molecule, bounces, hits another, and bounces again. Its path is a jagged, random zig-zag known as a “Random Walk.” The average distance a molecule travels between two successive collisions is called the Mean Free Path (l).
l = 1 / (√2 n π d²)
Where:
– n: Number density (molecules per unit volume)
– d: Diameter of the molecule
Physical Interpretation:
1. Size (d): If molecules are larger, they are bigger targets. They hit more often, so the free path ‘l’ decreases.
2. Density (n): If the gas is crowded (high density), collisions happen frequently, so ‘l’ decreases.
11. Comprehensive Practice Set
To master Physics, you must apply the concepts. Here is a curated set of problems ranging from conceptual checks to detailed calculations.
Part A: Very Short Answer Questions
- Q: Why is there no atmosphere on the Moon?
A: The value of root mean square velocity (vrms) of gas molecules on the Moon is greater than the Moon’s escape velocity. Hence, gases escape into space. - Q: Does the internal energy of an ideal gas change during isothermal expansion?
A: No. For an ideal gas, Internal Energy (U) depends only on Temperature. Since T is constant (Isothermal), U remains constant. - Q: Define Avogadro’s number.
A: It is the number of atoms in exactly 12g of C-12 isotope. Value = 6.022 × 1023 per mole.
Part B: Multiple Choice Questions (MCQ)
- The average kinetic energy of a gas molecule depends on:
(a) Pressure of the gas
(b) Volume of the gas
(c) Temperature of the gas
(d) Nature of the gasAnswer: (c). Remember the golden rule: E = 3/2 kBT. It depends only on T. - For an ideal gas, the ratio of Cp/Cv for a diatomic gas is:
(a) 1.67
(b) 1.40
(c) 1.33
(d) 1.50Answer: (b). For diatomic, f=5. Gamma = 1 + 2/5 = 1.4. - If the temperature of a gas is doubled, the root mean square (rms) speed becomes:
(a) 2 times
(b) 4 times
(c) √2 times
(d) HalfAnswer: (c). Vrms is proportional to √T. So if T doubles, speed becomes √2 times.
Part C: Conceptual Short Answers
- Q: When a gas is heated, its temperature increases. Explain this on the basis of kinetic theory.
Answer: Heat is a form of energy. When we supply heat to a gas, the molecules absorb this energy and start moving faster. Their translational kinetic energy increases. Since Kinetic Theory defines temperature as a measure of average kinetic energy ($T \propto v^2$), the increase in speed manifests as an increase in temperature.
- Q: Why do tyres burst in summer?
Answer: In summer, the high temperature causes the air molecules inside the tyre to move faster (higher kinetic energy). They hit the tyre walls harder and more frequently. This increases the internal pressure (Gay-Lussac’s Law: $P \propto T$). If this pressure exceeds the strength of the rubber, the tyre bursts.
Part D: Numerical Problems (Step-by-Step)
Problem 1: Calculating Kinetic Energy
Question: Calculate the average kinetic energy of an oxygen molecule at 27°C. (Boltzmann constant kB = 1.38 × 10-23 J/K).
Solution:
Step 1: Convert Temperature to Kelvin.
T = 27 + 273 = 300 K.
Step 2: Use the KE Formula.
E = (3/2) kB T
Step 3: Substitute values.
E = 1.5 × (1.38 × 10-23) × 300
E = 1.5 × 1.38 × 300 × 10-23
E = 6.21 × 10-21 Joules.
Problem 2: Degrees of Freedom & Energy
Question: A cylinder contains 1 mole of Helium and 1 mole of Nitrogen at the same temperature. Find the ratio of their total internal energies.
Solution:
Step 1: Identify gases and DoF (f).
Helium is Monatomic → f1 = 3.
Nitrogen is Diatomic → f2 = 5.
Step 2: Formula for Internal Energy.
U = (f/2) nRT.
Since n, R, and T are the same for both, U is directly proportional to f.
Step 3: Calculate Ratio.
UHe / UN2 = f1 / f2
Ratio = 3 : 5.
Problem 3: RMS Speed Calculation
Question: At what temperature will the rms speed of oxygen molecules be double their speed at 300 K?
Solution:
Step 1: Formula relation.
Vrms ∝ √T.
So, V1 / V2 = √(T1 / T2).
Step 2: Set condition.
We want V2 = 2V1.
V1 / 2V1 = √(300 / T2)
1/2 = √(300 / T2)
Step 3: Square both sides and solve.
1/4 = 300 / T2
T2 = 300 × 4 = 1200 K.
Problem 4: Mixture of Gases
Question: 1 mole of a monatomic gas is mixed with 1 mole of a diatomic gas. What is the value of γ for the mixture?
Solution:
Step 1: Calculate Total Internal Energy.
Umix = Umono + Udia
Umix = (3/2)RT + (5/2)RT = (8/2)RT = 4RT
Step 2: Effective Specific Heat (Cv-mix).
Umix = ntotal Cv-mix T
4RT = (1 + 1) Cv-mix T
4R = 2 Cv-mix → Cv-mix = 2R
Step 3: Effective Cp-mix.
Cp-mix = Cv-mix + R = 2R + R = 3R.
Step 4: Calculate γ.
γ = Cp / Cv = 3R / 2R = 1.5.

