
Start Chapter MCQ Quiz
Detailed Notes: Class 11 Physics Chapter 10 Thermal properties of Matter NCERT solutions
1. Introduction: Trusting the Physics, Not Your Feelings | Class 11 Physics Chapter 10 Thermal properties of Matter NCERT solutions
Hello students! Welcome to our deep dive into Chapter 10. I want you to take a moment and think about the room you are sitting in right now. Touch the wooden top of your desk. Now, touch the metal leg of the desk. Which one feels colder?
If you are like most people, you will say the metal leg feels colder. But here is the trick: they have both been in the same room for hours. They are actually at the exact same temperature! So why does the metal feel colder? It’s because metal steals heat from your hand faster than wood does. This simple observation teaches us a crucial lesson right at the start: Human senses are unreliable scientific instruments. We measure heat transfer, not temperature.
In this chapter, we are going to move away from subjective feelings like “hot” and “lukewarm” and move toward objective, mathematical definitions. We are going to study how matter behaves when it gets hot—how it expands, how it changes state, and how it passes energy along. This is the study of Thermal Properties of Matter.
2. Temperature and Heat: The Dynamic Duo
Let’s clear up the biggest confusion in thermal physics. In everyday language, we use “heat” and “temperature” interchangeably. In Physics, they are totally different concepts. Confusing them is like confusing “rain” with “a lake.” One is water falling (a process), and the other is water sitting there (a state).
2.1 Temperature (T)
Temperature is a measure of the “intensity” of heat. But what is it really? If we could zoom in with a super-microscope, we would see that every object is made of atoms vibrating, rotating, or flying around.
Definition: Temperature is the measure of the Average Kinetic Energy of the molecules of a substance.
- High Temperature: The molecules are moving violently.
- Low Temperature: The molecules are moving sluggishly.
- Absolute Zero (0 Kelvin): The theoretical point where all molecular motion ceases. It is the floor of the universe. You cannot go lower.
2.2 Heat (Q)
Heat is not something an object “has.” An object has Internal Energy. Heat is the energy in transit.
Definition: Heat is the form of energy that flows between two bodies or parts of a body because of a temperature difference.
The Water Level in the tanks represents Temperature.
The Water Flowing through the pipe represents Heat.
Water flows from High Level to Low Level, regardless of which tank has more total water. Similarly, Heat flows from High Temp to Low Temp, regardless of which object has more internal energy.
3. Measurement of Temperature (Thermometry)
Since we can’t see atoms vibrating, we need indirect ways to measure temperature. We use materials that change in a predictable way when heated. These changes are called Thermometric Properties.
- Liquid Thermometers: Mercury or Alcohol expands in a glass tube.
- Gas Thermometers: The pressure of a gas increases with heat (Constant Volume Gas Thermometer).
- Resistance Thermometers: The electrical resistance of a metal wire (like Platinum) increases as it gets hotter.
3.1 The Three Scales
Imagine we have a blank thermometer. We need to mark lines on it. This is called calibration.
1. Celsius Scale (°C): The everyday scale.
Lower Fixed Point (Ice Point): 0°C
Upper Fixed Point (Steam Point): 100°C
Number of divisions: 100
2. Fahrenheit Scale (°F): The clinical scale.
Lower Fixed Point: 32°F
Upper Fixed Point: 212°F
Number of divisions: 180
3. Kelvin Scale (K): The scientific scale.
Lower Fixed Point: 273.15 K
Upper Fixed Point: 373.15 K
Note: We never write degrees (°) with Kelvin.
3.2 Conversion Formulas
You must memorize these relations. They are the bread and butter of thermal physics problems.
T(K) = t(°C) + 273.15
t(°F) = (9/5)t(°C) + 32
t(°C) = (5/9)[t(°F) – 32]
3.3 The Ideal Gas Equation
For gases at low densities, pressure (P), volume (V), and temperature (T) are related by:
PV = nRT
Here, R is the Universal Gas Constant (8.31 J mol⁻¹ K⁻¹).
Crucial Note: Whenever you use this equation, T must be in Kelvin. If you put Celsius in here, your answer will be wrong, and the bridge you are designing might collapse!
4. Thermal Expansion
Have you ever noticed that railway tracks are laid with small gaps between them? Or that electric lines sag more in summer than in winter? This is due to thermal expansion.
Why does it happen?
When you heat a solid, the atoms vibrate with larger amplitudes. Think of a crowded dance floor. If everyone starts dancing wildly (higher temp), people need more space to avoid bumping into each other. The average distance between atoms increases, causing the whole material to expand.
4.1 Linear Expansion (Length)
This applies to rods, wires, and cables. The change in length is proportional to the original length and the rise in temperature.
ΔL = α L ΔT
Where α (Alpha) is the Coefficient of Linear Expansion.
Unit: K⁻¹ or °C⁻¹.
Example: Invar (a steel alloy) has a very small α, so it is used in clock pendulums to keep time accurate in summer and winter.
4.2 Area Expansion (Surface)
This applies to metal sheets or tiles. The area increases in two dimensions.
ΔA = β A ΔT
Where β (Beta) is the Coefficient of Area Expansion.
Relationship: β = 2α
4.3 Volume Expansion (3D)
This applies to blocks, spheres, and fluids (liquids/gases).
ΔV = γ V ΔT
Where γ (Gamma) is the Coefficient of Volume Expansion.
Relationship: γ = 3α
4.4 Anomalous Expansion of Water
Water is a rebel. While almost every other liquid contracts when cooled, water behaves strangely near its freezing point.
- As water cools from room temperature, it contracts (normal behavior).
- But from 4°C down to 0°C, water EXPANDS!
- This means water has its Maximum Density at 4°C.
Why is this important?
Because ice is less dense than water (due to this expansion), it floats. In winter, lakes freeze from the top down. The ice layer acts as a blanket, insulating the water below. The bottom of the lake remains at 4°C, allowing fish and aquatic life to survive the winter. If water behaved “normally,” lakes would freeze from the bottom up, killing everything!
5. Specific Heat Capacity (s)
Imagine you are at the beach on a sunny day. The sand burns your feet, but the water is cool. Why? The sun is beating down on both equally. The difference lies in their Specific Heat Capacity.
Definition: Specific Heat Capacity (s) is the amount of heat energy required to raise the temperature of 1 kg of a substance by 1°C (or 1 K).
Q = m s ΔT
- Sand: Low specific heat. It gets hot fast and cools down fast.
- Water: Extremely high specific heat (4186 J kg⁻¹ K⁻¹). It is very hard to heat up and very hard to cool down.
Application: Water is used as a coolant in car radiators because it can absorb a massive amount of heat from the engine without boiling over.
5.1 Calorimetry
This is the science of measuring heat. We use the Principle of Calorimetry (Conservation of Energy):
Heat Lost by Hot Body = Heat Gained by Cold Body
When solving these problems, always set up the equation:
m₁ s₁ (T₁ – T_final) = m₂ s₂ (T_final – T₂)
6. Change of State: The Hidden Heat
Matter can exist as Solid, Liquid, or Gas. Changing from one state to another requires energy, but surprisingly, it does not change the temperature.
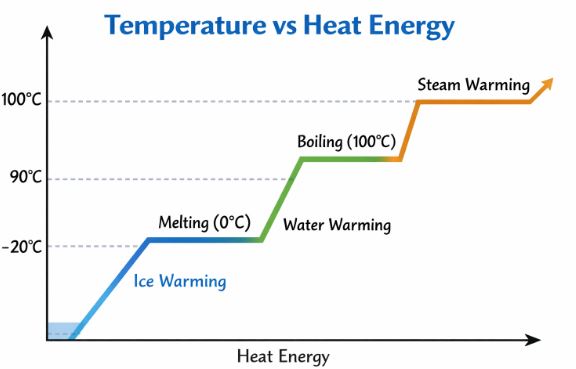
Figure 1: Heating Curve of Water. Note the flat regions (plateaus). This is where state change happens.
6.1 Latent Heat (L)
Look at the graph above. At 0°C, the graph goes flat. We are adding heat, but the temperature isn’t rising. Where is the energy going? It is being used to break the molecular bonds holding the solid together. This is called Latent Heat (Latent means “hidden”).
Q = m L
Types of Latent Heat:
- Latent Heat of Fusion (Lf): Solid to Liquid.
For water: Lf = 3.33 × 10⁵ J/kg. - Latent Heat of Vaporization (Lv): Liquid to Gas.
For water: Lv = 22.6 × 10⁵ J/kg.
Answer: Steam possesses “extra” heat energy in the form of Latent Heat of Vaporization. When steam touches your skin, it condenses into water and releases that massive amount of hidden energy (22.6 × 10⁵ J/kg) before it even starts cooling down like normal hot water.
7. Heat Transfer
Heat is restless. It always wants to move from a region of higher temperature to a region of lower temperature. There are three modes of transport.
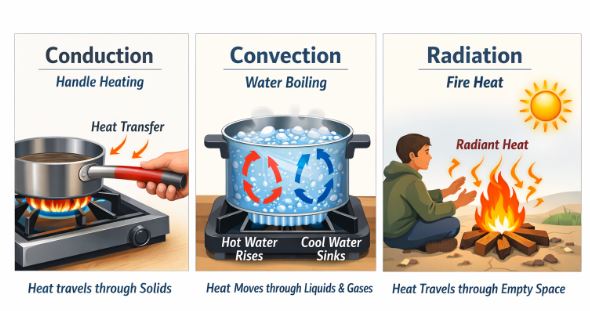
7.1 Conduction (Solids)
Imagine a line of students passing a basketball from one end to the other without moving from their spots. This is conduction.
At the hot end, atoms vibrate violently and collide with their neighbors, transferring energy. In metals, “free electrons” zip through the material, carrying energy very fast, which makes metals good conductors.
H = KA (T₁ – T₂) / L
Where K is Thermal Conductivity. High K means a good conductor (Copper, Aluminum). Low K means an insulator (Wood, Styrofoam).
7.2 Convection (Fluids)
Imagine the students now running with the basketball to the other side. This is convection.
It involves the actual movement of matter. When water at the bottom of a pot heats up, it expands, becomes less dense (lighter), and rises. Cold, dense water sinks to take its place. This creates a Convection Current.
Real World Example: Sea Breeze
During the day, land heats up faster than the sea. Air over land rises (Low Pressure). Cool air from the sea rushes in to fill the gap. That nice breeze you feel at the beach is convection in action!
7.3 Radiation (Vacuum)
How does heat from the Sun reach Earth? There is no air in space for conduction or convection. It travels as Electromagnetic Waves (Infrared). This is Radiation.
Stefan-Boltzmann Law:
The energy radiated per second by a black body is proportional to the fourth power of its absolute temperature.
E = σ T⁴
This means if you double the temperature of an object (2T), the radiated energy increases by 16 times (2⁴)!
8. Newton’s Law of Cooling
Sir Isaac Newton observed that a hot body cools down faster when it is very hot, and slows down as it approaches room temperature.
Statement: The rate of loss of heat is directly proportional to the temperature difference between the body and the surroundings.
-dQ/dt = K (T_body – T_surroundings)
The cooling curve is not a straight line; it is an exponential decay curve.
9. Comprehensive Solved Examples
Physics is best learned by doing. Let’s tackle some problems ranging from easy to “exam level.”
Example 1: Temperature Conversion
Question: A faulty thermometer reads 5°C in melting ice and 95°C in steam. If this thermometer reads 59°C, what is the correct temperature in Celsius?
Teacher’s Solution:
This is a classic “calibration” problem. We compare the “ratios” of the scales.
Formula: (Reading – Lower Fixed Point) / (Upper Fixed Point – Lower Fixed Point) = Constant
Let C be the correct temperature.
Step 1: Setup for Correct Scale
(C – 0) / (100 – 0) = C / 100
Step 2: Setup for Faulty Scale
(59 – 5) / (95 – 5) = 54 / 90
Step 3: Equate them
C / 100 = 54 / 90
C = (54 / 90) × 100
C = 0.6 × 100 = 60°C
Answer: The correct temperature is 60°C.
Example 2: Thermal Expansion
Question: A steel bridge is 200 m long at 20°C. If the temperature drops to -10°C in winter and rises to 40°C in summer, what is the difference in length between the coldest and hottest days? (α for steel = 1.2 × 10⁻⁵ K⁻¹)
Teacher’s Solution:
We need to find the change in length over the total temperature range.
Step 1: Calculate Total Temperature Difference (ΔT)
Max Temp = 40°C
Min Temp = -10°C
ΔT = 40 – (-10) = 50°C (or 50 K)
Step 2: Apply Formula
ΔL = α L ΔT
ΔL = (1.2 × 10⁻⁵) × 200 × 50
Step 3: Calculate
ΔL = 1.2 × 10⁻⁵ × 10000
ΔL = 1.2 × 10⁻¹ = 0.12 meters or 12 cm.
Answer: The bridge expands and contracts by 12 cm. This is why engineers put expansion joints (rollers) at the ends of bridges!
Example 3: Calorimetry (The Coffee Problem)
Question: You pour 0.2 kg of hot coffee at 90°C into a 0.3 kg ceramic mug at 20°C. What is the final temperature? (Assume specific heat of coffee = 4000 J/kg·K and mug = 1000 J/kg·K). Ignore heat loss to air.
Teacher’s Solution:
Let the final equilibrium temperature be T.
Heat Lost by Coffee = Heat Gained by Mug.
Step 1: Heat Lost by Coffee
Q_lost = m × s × ΔT
Q_lost = 0.2 × 4000 × (90 – T)
Q_lost = 800 (90 – T)
Step 2: Heat Gained by Mug
Q_gain = m × s × ΔT
Q_gain = 0.3 × 1000 × (T – 20)
Q_gain = 300 (T – 20)
Step 3: Equate and Solve
800 (90 – T) = 300 (T – 20)
Divide both sides by 100 to simplify:
8 (90 – T) = 3 (T – 20)
720 – 8T = 3T – 60
720 + 60 = 3T + 8T
780 = 11T
T = 780 / 11 ≈ 70.9°C
Answer: The final temperature is roughly 71°C.
Example 4: The Iceberg Challenge (Latent Heat)
Question: How much heat is required to convert 10 g of ice at -5°C into steam at 100°C?
Data: s_ice = 2100 J/kg·K, s_water = 4186 J/kg·K, L_fusion = 3.33×10⁵ J/kg, L_vapor = 22.6×10⁵ J/kg.
Teacher’s Solution:
This is a multi-step journey. We cannot do this in one calculation. We must break it down into stages.
Step 1: Heat Ice from -5°C to 0°C (Q1)
m = 10g = 0.01 kg.
Q1 = m s_ice ΔT = 0.01 × 2100 × 5 = 105 J
Step 2: Melt Ice at 0°C to Water at 0°C (Q2)
This uses Latent Heat.
Q2 = m L_f = 0.01 × 3.33×10⁵ = 3330 J
Step 3: Heat Water from 0°C to 100°C (Q3)
Q3 = m s_water ΔT = 0.01 × 4186 × 100 = 4186 J
Step 4: Boil Water at 100°C to Steam at 100°C (Q4)
Q4 = m L_v = 0.01 × 22.6×10⁵ = 22600 J
Step 5: Total Heat
Total = 105 + 3330 + 4186 + 22600 = 30,221 Joules (approx 30.2 kJ).
Observation: Notice that Q4 (boiling) is massive compared to the others. Making steam takes a LOT of energy!
10. Summary & Key Takeaways
We have covered a lot of ground today! Let’s summarize the key points to stick on your mental wall:
- Temperature ≠ Heat: Temperature is the average KE (State), Heat is the flow of energy (Process).
- Kelvin is King: Always convert Celsius to Kelvin in Gas Laws (T = t + 273).
- Expansion: Things expand when heated (mostly). Water contracts from 0-4°C (Anomaly).
- Specific Heat (s): Water has high thermal inertia; sand has low.
- Latent Heat (L): Energy used to break bonds during state change, not to raise temperature.
- Heat Transfer: Conduction (solids), Convection (fluids), Radiation (vacuum).
Keep practicing the numericals, especially the calorimetry ones, as they require careful accounting of energy. Physics is logical—if your energy balance doesn’t match, check your signs! See you in the next lecture!
Read Also:
Class-11 Chapter 9- Mechanical Properties of Fluids
For more check official website of
NCERT
