1. Introduction: The Engine of the Universe | Class 11 Physics Chapter 11 Thermodynamics Notes
Good morning, class! Today, we are opening the door to one of the most powerful and influential branches of science: Thermodynamics. While our previous lessons in Mechanics dealt with visible objects like rolling balls, falling apples, and moving cars, Thermodynamics dives deeper. It deals with the invisible energy that drives those movements. It is the study of Heat, Work, and the Internal Energy of systems.
The name itself gives us a clue: Thermo means “Heat” and Dynamics means “Power” or “Motion”. Historically, this science wasn’t born in a quiet laboratory; it was born in the noisy, soot-filled factories of the Industrial Revolution. Engineers were desperate to answer one question: “How do we get more work out of a lump of coal?” They wanted to build better steam engines. From those practical questions, we discovered universal laws that govern everything from the engine of a Ferrari to the fusion reactions in the Sun, and even the digestion of the breakfast you ate this morning.
In Chapter 10, we looked at molecular details—why water expands when it freezes, or how molecules vibrate.
In Chapter 11 (Thermodynamics), we zoom out. We stop caring about individual molecules. We look at the “Big Picture” or Macroscopic Variables like Pressure, Volume, and Temperature. We treat the gas as a single “System” and study how it exchanges energy.
2. Fundamental Concepts: Speaking the Language
Before we can master the laws, we must agree on our vocabulary. Thermodynamics is very precise about definitions.
2.1 System and Surroundings
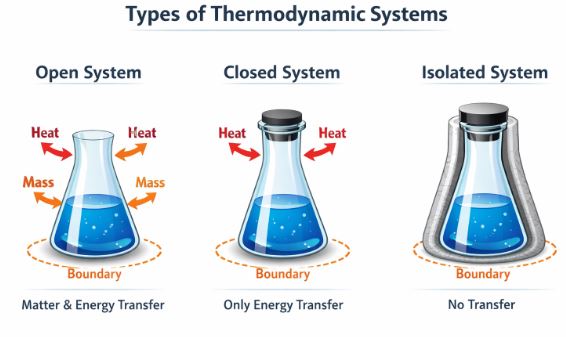
Imagine you are studying a balloon filled with helium.
- The System: The specific part of the universe under study. In this case, the helium gas inside the balloon.
- The Surroundings: Everything else in the universe. The rubber of the balloon, the air in the room, you, and the rest of the world.
- The Boundary: The surface (real or imaginary) that separates the system from the surroundings. The nature of this boundary dictates what can happen.
2.2 Types of Walls (Boundaries)
The wall of a container determines if heat can enter or leave.
- Adiabatic Wall: Think of this as a “perfect insulator.” It allows NO heat transfer. If a system is enclosed in adiabatic walls, its temperature can change only if we do work on it (like compressing it), not by heating it.
Example: A high-quality thermos flask or Styrofoam cup. - Diathermic Wall: Think of this as a “perfect conductor.” It allows heat to flow freely. If two systems are separated by a diathermic wall, they will exchange heat until they reach the same temperature.
Example: A thin copper sheet or an aluminum kettle.
2.3 Thermodynamic State Variables
To describe the condition (or “state”) of a system, we use measurable macroscopic quantities. These are divided into two categories:
| Extensive Variables | Intensive Variables |
|---|---|
| Depend on the size or mass of the system. | Independent of the size or mass of the system. |
| Test: If you divide the system in half, these values get halved. | Test: If you divide the system in half, these values remain unchanged. |
| Examples: Volume (V), Mass (m), Internal Energy (U). | Examples: Pressure (P), Temperature (T), Density. |
3. The Zeroth Law of Thermodynamics
The Zeroth Law of Thermodynamics
It sounds strange to start counting at zero, doesn’t it? This law was formulated by Ralph Fowler in 1931, long after the First and Second Laws were well-established. However, physicists realized that this concept was logically fundamental to the others, so they placed it at the beginning.
3.1 The Statement
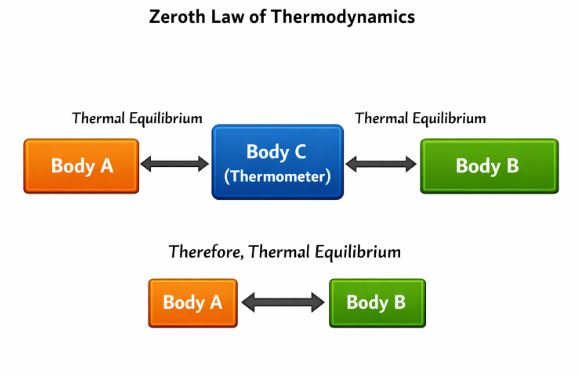
“If two systems A and B are continuously in thermal equilibrium with a third system C, then A and B are also in thermal equilibrium with each other.”
3.2 Why is this significant?
This law gives us the scientific definition of Temperature.
Imagine System C is a Thermometer.
1. You place the thermometer (C) in contact with a cup of coffee (A). The reading stops changing at 70°C.
2. You place the same thermometer (C) in contact with a beaker of hot water (B). The reading stops changing at 70°C.
3. According to the Zeroth Law, we can now guarantee that if you mix A and B, there will be no net heat flow between them. They are in Thermal Equilibrium.
Therefore, Temperature is simply the physical property that determines whether a system is in equilibrium with another.
4. The First Law of Thermodynamics
This is the bedrock of physics. It is essentially the Law of Conservation of Energy applied to thermodynamic systems. It tells us that energy cannot be created or destroyed, only transformed.
4.1 Internal Energy (U)
Every substance possesses a hidden store of energy called Internal Energy. It is the sum of two things:
1. Kinetic Energy: The random motion of molecules (translation, rotation, vibration). This depends directly on Temperature.
2. Potential Energy: The energy due to intermolecular forces (attraction/repulsion). This depends on Volume (spacing between molecules).
Important for Ideal Gases: In an Ideal Gas, we assume there are no intermolecular forces (no Potential Energy). Therefore, the Internal Energy of an ideal gas depends ONLY on Temperature.
4.2 The Mathematical Form
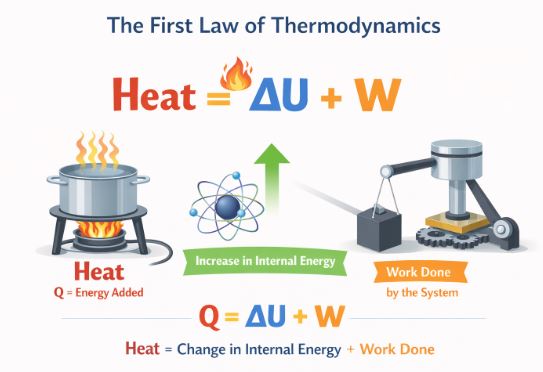
ΔQ = ΔU + ΔW
This equation tells a simple accounting story. When you supply Heat energy (ΔQ) to a system, that energy can go to two places:
1. Increasing the system’s own Internal Energy (ΔU) (making it hotter).
2. Doing mechanical Work (ΔW) on the surroundings (like pushing a piston).
4.3 The Sign Convention (Crucial!)
If you get the signs wrong, your entire calculation will be wrong. In Physics (IUPAC convention for Heat, Mechanical convention for Work), we follow this:
- Heat (ΔQ):
- Heat given TO the system: Positive (+)
- Heat taken FROM the system: Negative (-)
- Work (ΔW):
- Work done BY the system (Expansion): Positive (+). (The gas pushes the piston up).
- Work done ON the system (Compression): Negative (-). (You push the piston down).
- Internal Energy (ΔU):
- Temperature Increases: Positive (+)
- Temperature Decreases: Negative (-)
5. Specific Heat Capacities of Gases
In solids and liquids, heating is straightforward. But gases are compressible, which makes things complicated. The amount of heat required to raise the temperature of a gas depends heavily on how the gas is confined.
5.1 Cv (Molar Specific Heat at Constant Volume)
Imagine a gas trapped in a rigid steel box. It cannot expand.
Since Volume is constant, Work Done (ΔW = PΔV) is Zero.
From the First Law: ΔQ = ΔU.
This means 100% of the heat you supply goes directly into raising the temperature.
Formula: (ΔQ)v = n Cv ΔT
5.2 Cp (Molar Specific Heat at Constant Pressure)
Now, imagine the gas in a cylinder with a movable piston. As you heat it, the gas expands to keep the pressure constant.
The gas does Work (ΔW) by pushing the piston.
From the First Law: ΔQ = ΔU + ΔW.
Here, the heat you supply must do two jobs: raise the temperature (ΔU) AND pay for the work done (ΔW).
Formula: (ΔQ)p = n Cp ΔT
This is a favorite exam question! At constant pressure, you need “extra” heat to compensate for the energy lost in doing expansion work. At constant volume, no work is done, so you need less heat for the same temperature rise. Hence, Cp is always greater than Cv.
5.3 Mayer’s Relation
For one mole of an ideal gas, the relationship is precise:
Cp – Cv = R
Where R is the Universal Gas Constant (8.314 J mol-1 K-1).
We also define the Ratio of Specific Heats (γ):
γ = Cp / Cv
The value of γ depends on the atomicity of the gas:
– Monatomic (He, Ar): γ = 1.67
– Diatomic (N2, O2): γ = 1.4
– Polyatomic (CO2): γ = 1.33
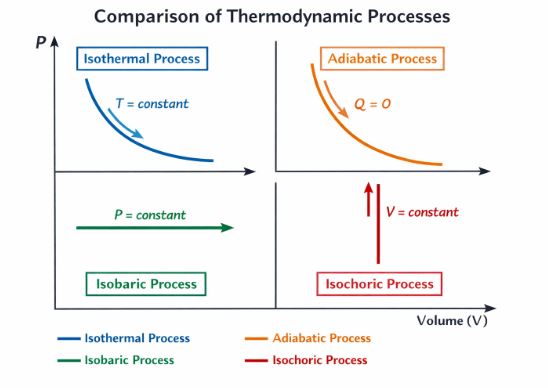
6. Thermodynamic Processes
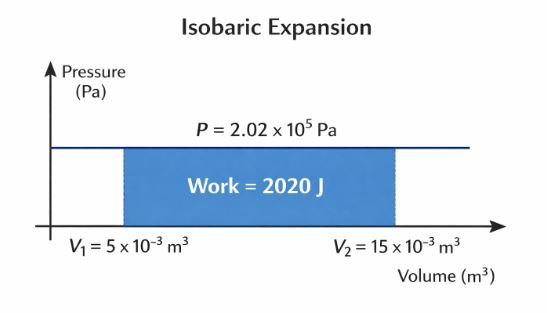
A “Process” describes how a system changes from State 1 (P1, V1, T1) to State 2 (P2, V2, T2). We visualize these on a P-V Diagram (Indicator Diagram). The most important rule to remember is: The Area under the P-V Curve equals the Work Done.