
Class 9 Atoms and Molecules Notes – Chapter 3 Science
1. Introduction to Class 9 Atoms and Molecules
Have you ever wondered what everything around you is made of? Your book, the air, your food, even you? For thousands of years, people have been asking this question. Around 500 BC, an Indian philosopher named Maharishi Kanad suggested that if you keep dividing matter (which he called padarth), you would eventually reach a tiny, indivisible particle he called parmanu. Around the same time, Greek philosophers like Democritus had a similar idea and called these fundamental particles atoms, which means “indivisible.”
These were just ideas for a long time. It wasn’t until scientists started doing experiments that we began to truly understand the world of atoms.
2. The Rules of the Game: Laws of Chemical Combination
In the late 18th century, scientists like Antoine Lavoisier and Joseph L. Proust established two fundamental laws that govern how elements combine to form new substances. These laws are the bedrock of modern chemistry.
2.1 The Law of Conservation of Mass
This law is quite simple but very powerful. It states that: Mass can neither be created nor destroyed in a chemical reaction.
In chemistry, the substances you start with are called reactants, and the new substances you form are called products. This law says: Total Mass of Reactants = Total Mass of Products. This is why, in a lab experiment, if you mix two solutions in a sealed flask and weigh it before and after the reaction, the mass will remain exactly the same.
2.2 The Law of Constant Proportions (or Definite Proportions)
This law, stated by Proust, tells us about the recipe for making compounds. It states that: In a chemical substance, the elements are always present in definite proportions by mass.
Think of water (H₂O). No matter where you get it from, the ratio of the mass of hydrogen to the mass of oxygen is always 1:8. If you have 9 grams of water, it will always be made of 1 g of hydrogen and 8 g of oxygen. This fixed recipe is what makes a compound a pure substance.
3. Dalton’s Atomic Theory: Putting it all Together
A British chemist named John Dalton came up with the first scientific theory about atoms in 1808. His theory was brilliant because it explained the two laws we just discussed.

Dalton was the first to use symbols to represent elements in a scientific context.
Here are the main points (postulates) of Dalton’s Atomic Theory:
- All matter is made of tiny particles called atoms.
- Atoms are indivisible particles, which cannot be created or destroyed in a chemical reaction. (This explains the Law of Conservation of Mass).
- Atoms of a given element are identical in mass and chemical properties.
- Atoms of different elements have different masses and chemical properties.
- Atoms combine in the ratio of small whole numbers to form compounds.
- The relative number and kinds of atoms are constant in a given compound. (This explains the Law of Constant Proportions).
(Note: We now know that atoms are not truly “indivisible”—they are made of even smaller particles like protons, neutrons, and electrons. However, Dalton’s theory was a revolutionary step forward!)
4. What is an Atom?
An atom is the smallest particle of an element that can take part in a chemical reaction. Think of atoms as the LEGO bricks of the universe. Atoms are incredibly small, their radius is measured in nanometers (nm), where 1 nm = 10⁻⁹ meters.
4.1 Symbols of Atoms
Today, we use a system approved by the IUPAC (International Union of Pure and Applied Chemistry). The symbol is usually the first one or two letters of the element’s name. The first letter is always uppercase, and the second is always lowercase (e.g., Al, not AL). Some symbols come from Latin or Greek names, like Fe for iron (from ferrum).
5. What is Atomic Mass?
Since atoms are so light, scientists use relative atomic mass. They compare the mass of an atom to a standard atom, which is Carbon-12. The atomic mass unit (u) is defined as: Exactly 1/12th the mass of one atom of Carbon-12. So, when we say the atomic mass of Hydrogen is 1 u, we mean it has a mass equal to 1/12th of a Carbon-12 atom.
6. Molecules and Ions: Atoms in Groups
Atoms rarely exist alone. They team up to form larger particles called molecules and ions.
6.1 What is a Molecule?
A molecule is a group of two or more atoms chemically bonded together. The number of atoms in a molecule is its atomicity (e.g., Helium He is monoatomic, Oxygen O₂ is diatomic, Ozone O₃ is polyatomic).
6.2 What is an Ion?
An ion is an atom or a group of atoms that has an electrical charge from losing or gaining electrons. A positively charged ion is a cation (e.g., Na⁺), and a negatively charged ion is an anion (e.g., Cl⁻). A group of atoms with a charge is a polyatomic ion (e.g., NO₃⁻).
7. Writing Chemical Formulae
A chemical formula is a shorthand way of representing a compound. To write one, you need to know the valency of the elements, which is their combining power.
7.1 The Criss-Cross Method
- Write the symbols of the elements/ions side-by-side.
- Write the valency (or charge) above each symbol.
- Criss-cross the valencies; they become the subscript for the other atom.
- Simplify the ratio and omit subscripts of 1. Use brackets for polyatomic ions if their subscript is greater than 1.
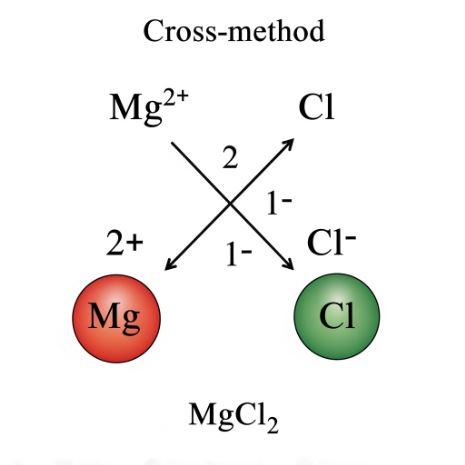
The criss-cross method is a simple way to determine the chemical formula of an ionic compound.
8. Molecular Mass and Formula Unit Mass
Molecular Mass is the sum of the atomic masses of all atoms in one molecule (e.g., H₂O = (2×1u) + (1×16u) = 18 u). The term Formula Unit Mass is used for ionic compounds and is calculated the same way (e.g., NaCl = (1×23u) + (1×35.5u) = 58.5 u).
9. Extensive Practice Set
Part A: Multiple-Choice Questions (MCQs)
- Which postulate of Dalton’s Atomic Theory explains the Law of Conservation of Mass?
a) Atoms of a given element are identical in mass.
b) Atoms combine in the ratio of small whole numbers.
c) Atoms are indivisible and cannot be created or destroyed.
d) The relative number and kinds of atoms are constant in a compound. - The mass ratio of Nitrogen to Hydrogen in ammonia is always 14:3. This observation is explained by:
a) The Law of Conservation of Mass
b) The Law of Constant Proportions
c) The Law of Multiple Proportions
d) Dalton’s Law of Partial Pressures - What is the correct chemical formula for Aluminium Sulphate? (Valency of Al = 3+, Sulphate ion = SO₄²⁻)
a) AlSO₄
b) Al₂(SO₄)₃
c) Al₃(SO₄)₂
d) Al₂SO₄ - The atomicity of Ozone (O₃), Phosphorus (P₄), and Helium (He) are respectively:
a) 3, 4, and 1
b) Diatomic, Polyatomic, and Monoatomic
c) 1, 4, and 3
d) Polyatomic, Diatomic, and Monoatomic - The modern atomic mass unit (u) is defined based on which standard?
a) 1/16th the mass of an Oxygen-16 atom
b) The mass of a Hydrogen-1 atom
c) 1/12th the mass of a Carbon-12 atom
d) 1/14th the mass of a Nitrogen-14 atom
Part B: Short Answer Questions
- Define an ion. What is the difference between a cation and an anion? Give one example of each.
- Calculate the molecular mass of Sulphuric Acid (H₂SO₄). (Atomic masses: H=1 u, S=32 u, O=16 u)
- Explain the “criss-cross” method for writing a chemical formula using the example of Calcium Nitrate (Ca²⁺ and NO₃⁻).
- State the Law of Conservation of Mass. If 12 g of carbon reacts completely with 32 g of oxygen, what mass of carbon dioxide will be formed?
- What do the following symbols/formulae represent?
a) Co vs. CO
b) H₂ vs. 2H
c) O²⁻
Part C: Long Answer Questions
- List and explain any four postulates of Dalton’s Atomic Theory. For two of these postulates, explain how they support the laws of chemical combination.
- What is valency? Write the chemical formulae for the following compounds:
a) Sodium Oxide
b) Magnesium Hydroxide
c) Iron(III) Chloride
d) Ammonium Phosphate - Differentiate between a molecule of an element and a molecule of a compound. Provide two examples for each and also mention their atomicity.
- An element ‘X’ has a valency of 3. An element ‘Y’ has a valency of 2.
a) What is the chemical formula of the compound formed between X and Y?
b) If ‘X’ is a metal and ‘Y’ is a non-metal, what type of compound is it (ionic/covalent)? What are the ions that would be formed? - A student performs an experiment where 9.0 g of water is decomposed by passing electricity. It produces 1.0 g of Hydrogen gas and 8.0 g of Oxygen gas. Which law of chemical combination does this experiment verify? State the law and explain how the results support it. What would be the mass of oxygen required to react completely with 4 g of hydrogen gas?
- Students can use these Class 9 Atoms and Molecules Notes for quick revision before exams
Read Also:
Class 9 Is Matter Around Us Pure?
Chapter Quiz
Frequently Asked Questions – Class 9 Atoms and Molecules Notes
What are atoms and molecules in Class 9 Science?
In Class 9 Science, atoms are the smallest particles of matter that take part in chemical reactions, while molecules are formed when two or more atoms chemically combine. These Class 9 Atoms and Molecules Notes explain the concept in a simple and exam-oriented way.
Why are Class 9 Atoms and Molecules Notes important for exams?
Class 9 Atoms and Molecules Notes are important because this chapter forms the base of chemistry. These notes help students understand laws of chemical combination, atomic mass, molecular mass, and mole concept clearly, which are frequently asked in exams.
Which laws are covered in Class 9 Atoms and Molecules Notes?
Class 9 Atoms and Molecules Notes cover important laws such as the Law of Conservation of Mass and the Law of Constant Proportions. These laws explain how atoms and molecules combine during chemical reactions.
What is atomic mass and molecular mass in Class 9 Atoms and Molecules?
In Class 9 Atoms and Molecules Notes, atomic mass refers to the mass of a single atom compared to carbon-12, while molecular mass is the sum of atomic masses of all atoms present in a molecule.
How do Class 9 Atoms and Molecules Notes help in numerical problems?
Class 9 Atoms and Molecules Notes explain numerical problems based on mole concept, atomic mass, and molecular mass step by step, making calculations easy for students during exams.
