
Start Chapter MCQ Quiz
Detailed Notes: Class 11 Physics Chapter 9 Mechanical Properties of fluids Notes
1. Introduction: The World of Flow | Class 11 Physics Chapter 9 Mechanical Properties of fluids Notes
Hello students! Welcome to Chapter 9. Up until now, we have dealt with solids—rigid bodies like blocks, cars, and planets. Solids are stubborn; they resist deformation. But today, we enter the world of Fluids.
What exactly is a Fluid?
In simple terms, a fluid is any substance that can flow. This means it has no fixed shape and takes the shape of its container. This category includes both Liquids and Gases.
1.1 The Molecular Difference
Why do solids hold their shape while fluids flow? It comes down to the relationship between the molecules.
- Solids: The molecules are tightly packed in a rigid lattice structure. They have strong intermolecular forces that hold them in place. If you push one, the whole structure moves.
- Fluids: The intermolecular forces are weaker. The molecules are free to slide past one another.
- Liquids: Molecules are close together (like a crowd at a concert), so they are very hard to compress. They have a definite volume but no definite shape.
- Gases: Molecules are far apart (like players on a football field). They have very weak forces and high kinetic energy. They expand to fill any volume available and are highly compressible.
1.2 Shear Stress in Fluids
Here is the technical definition of a fluid: A substance that deforms continuously under shear stress.
Imagine a block of jelly (solid). If you push the top sideways, it deforms a bit and stops. It resists the shear stress.
Now imagine water. If you push the top layer of water sideways, it doesn’t stop deforming. It flows. No matter how small the force is, a fluid will eventually give way and flow.
2. Pressure: The Force of Fluids
When you dive into a swimming pool, you feel a weight pressing on your ears. That is the weight of the water pushing on you. In fluids, we talk about Pressure rather than just Force.
Definition: Pressure is the Force acting perpendicular (normal) to a surface per unit Area.
Formula: P = F / A
- F: Normal Force (Thrust).
- A: Area over which force is applied.
SI Unit: Pascal (Pa).
1 Pa = 1 Newton per square meter (N/m²).
Since 1 Pa is very small (like the weight of a sheet of paper on a table), we often use Atmosphere (atm) or Bar.
1 atm = 1.013 × 10⁵ Pa.
Is Pressure a Vector?
No! Pressure is a Scalar Quantity.
“But sir, force has a direction!” you might ask.
True, but pressure acts in all directions equally. If you submerge a balloon in water, it gets squeezed from the top, bottom, left, and right. Since it has no unique direction, it is a scalar.
2.1 Density (ρ)
Before we go deeper, we need to understand Density. It measures how “packed” the matter is.
Density (ρ) = Mass (m) / Volume (V)
– Water has a density of 1000 kg/m³.
– Mercury is very dense: 13,600 kg/m³.
– Air is light: 1.29 kg/m³.
3. Pascal’s Law: The Magic Multiplier
In 1653, the French scientist Blaise Pascal discovered something amazing about confined fluids.
Pascal’s Law States: “If gravity is ignored, the pressure applied to an enclosed fluid is transmitted undiminished to every portion of the fluid and the walls of the containing vessel.”
This means if you squeeze a toothpaste tube at the bottom, the pressure travels all the way to the top and pushes the paste out. The pressure doesn’t get “used up” along the way.
3.1 Application: The Hydraulic Lift
We use this law to lift heavy cars with very little effort. Let’s see the math behind this “magic.”
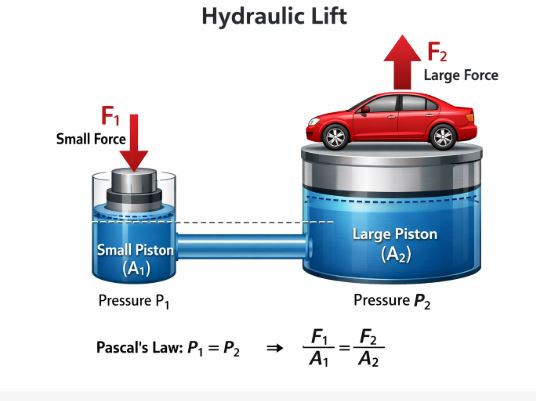
Figure 1: The Hydraulic Lift. A small push creates a giant lift.
Imagine two cylinders connected by a pipe filled with oil.
– Input Cylinder: Narrow, Area A₁.
– Output Cylinder: Wide, Area A₂.
You apply a small force F₁ on the small piston.
Pressure created: P = F₁ / A₁.
According to Pascal, this same pressure P travels to the large piston.
Force exerted by large piston: F₂ = P × A₂.
Substitute P: F₂ = (F₁ / A₁) × A₂
F₂ = F₁ × (A₂ / A₁).
Since A₂ is much larger than A₁, F₂ is much larger than F₁.
Example: If A₂ is 100 times bigger than A₁, you can lift a 1000 kg car by applying a force equal to just 10 kg! This is called Force Multiplication.
3.2 Hydraulic Brakes
The same principle stops your car. When you press the brake pedal (small piston), the pressure travels through the brake fluid to the wheels (large pistons), pushing the brake pads against the discs with massive force to stop the spinning wheels.
4. Variation of Pressure with Depth
If you dive deep into the ocean, the weight of all the water above you pushes down on you. This is why submarines have thick steel hulls.
Consider a fluid at rest. Imagine a small cylinder of liquid inside the container at depth h.
The force pushing down on the base of this cylinder is the weight of the liquid column above it.
Weight = Mass × g = (Volume × Density) × g = (Area × h × ρ) × g.
Pressure = Force / Area = (A h ρ g) / A.
P = hρg.
The Total Pressure Formula:
If the surface of the liquid is open to the air, we must add the weight of the air (Atmospheric Pressure) too.
P_total = P_atmosphere + ρgh
– P_total is called Absolute Pressure.
– ρgh is called Gauge Pressure (pressure due to the liquid only).
The Hydrostatic Paradox:
Does the shape of the container matter? If you have a thin tube and a wide bucket, both filled to height ‘h’, is the pressure at the bottom different?
NO! Pressure depends only on height ‘h’, not the volume or shape. The pressure at the bottom of a 1-meter straw filled with water is the same as the pressure at the bottom of a 1-meter deep swimming pool!
5. Atmospheric Pressure and Its Measurement
We live at the bottom of an ocean of air called the atmosphere. This air has mass, so it has weight. The weight of this air pressing down on us is Atmospheric Pressure (Pₐ).
At sea level, Pₐ = 1.013 × 10⁵ Pa.
This is a huge amount! It’s equivalent to an elephant standing on your head. Why don’t we get crushed? Because the blood pressure inside our bodies pushes outwards with roughly the same force, balancing it.
5.1 Torricelli’s Experiment (The Barometer)
Evangelista Torricelli invented the Barometer to measure this pressure.
He took a long glass tube closed at one end, filled it with mercury, and inverted it into a bowl of mercury.
The mercury level dropped a bit but stopped at a height of 76 cm (760 mm).
Why didn’t it all fall out? Because the atmospheric air pushes down on the mercury in the bowl, forcing the mercury up the tube. The weight of the 76 cm mercury column exactly balances the weight of the atmosphere.
Why Mercury?
Mercury is very dense (13.6 times water). If we used water, the barometer tube would have to be 10.3 meters tall! Mercury allows us to keep the instrument compact.
6. Fluid Dynamics: Fluids in Motion
So far, we looked at fluids at rest (Hydrostatics). Now let’s let the water flow! This branch is Hydrodynamics.
6.1 Types of Flow
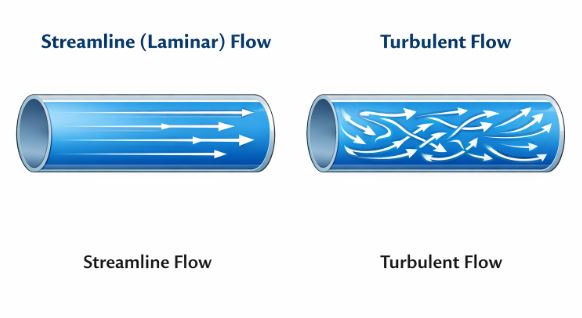
1. Streamline (Laminar) Flow
Imagine soldiers marching in a parade. Everyone follows the exact path of the person in front. This is streamline flow. It occurs at low speeds. The layers of fluid slide smoothly over each other.
2. Turbulent Flow
Imagine a crowd rushing out of a stadium in panic. It’s chaotic. Particles mix, swirl, and form eddies (whirlpools). This happens at high speeds, like a fast river or smoke from a chimney.
Critical Velocity & Reynolds Number:
There is a specific speed limit called Critical Velocity. Below this, flow is streamline; above it, flow becomes turbulent.
Engineers use a dimensionless number called Reynolds Number (Re) to predict flow type.
– Re < 1000: Laminar
– Re > 2000: Turbulent
6.2 Equation of Continuity
This is simply the Conservation of Mass applied to fluids.
Imagine water flowing through a pipe that has wide and narrow sections. Since water cannot be compressed or destroyed, the amount of water entering one end must equal the amount leaving the other end every second.
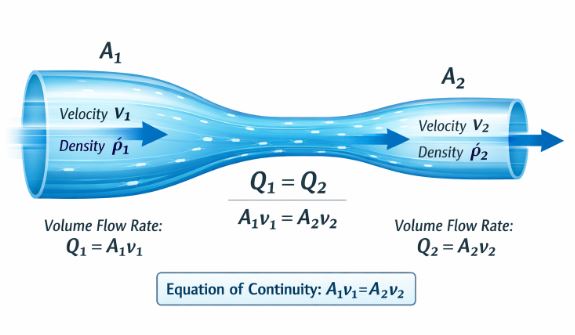
Figure 2: Continuity. As area shrinks, speed grows.
Rate of Flow = Area × Velocity
A₁ v₁ = A₂ v₂ = Constant
This means Area and Velocity are inversely proportional.
– Wide Pipe (Large A) -> Slow Speed (Low v).
– Narrow Pipe (Small A) -> Fast Speed (High v).
Real Life: When you water plants with a hose, you put your thumb over the opening to make the hole smaller. The water shoots out faster and reaches further!
7. Bernoulli’s Principle: The Physics of Flight
Daniel Bernoulli, a Swiss mathematician, gave us the Conservation of Energy for flowing fluids.
For a streamline flow of an ideal fluid, the total energy (Pressure Energy + Kinetic Energy + Potential Energy) per unit volume remains constant.
Formula: P + ½ρv² + ρgh = Constant
The Practical Insight:
If a fluid flows horizontally (height h is constant), then:
P + ½ρv² = Constant
This means if Velocity (v) increases, Pressure (P) must decrease to keep the sum constant.
High Speed = Low Pressure.
Low Speed = High Pressure.
Applications of Bernoulli’s Principle
This principle explains many “magic” tricks of nature.
1. Dynamic Lift (Why Airplanes Fly)
An airplane wing (aerofoil) is curved on top and flat on the bottom. When the plane moves, air has to travel longer over the curved top surface, so it speeds up.
– Top: High Air Speed -> Low Pressure.
– Bottom: Low Air Speed -> High Pressure.
The high pressure from below pushes the wing UP. This upward force is called Lift.
2. The Magnus Effect (Spinning Ball)
In cricket or football, a spinning ball curves in the air (Swing).
As the ball spins, it drags air with it. On one side, the spin helps the wind (High Speed -> Low P). On the other side, the spin fights the wind (Low Speed -> High P). The ball gets pushed towards the Low Pressure side, making it curve.
3. Blowing off Roofs
During a storm, high-speed wind blows over the roof. This creates extremely Low Pressure above the roof. The air inside the room is still (High Pressure). This pressure difference lifts the roof up and blows it away!
8. Viscosity: The Fluid Friction
Try pouring water and then try pouring honey. Honey is slow. Why? Because it is Viscous.
Viscosity is the internal friction between the layers of a fluid.
When a fluid flows, it flows in layers. The layer touching the pipe is stationary. The center layer moves fastest. The layers drag against each other.
Coefficient of Viscosity (η – eta):
This measures how “thick” or sticky a fluid is.
– High η: Honey, Glycerine, Oil.
– Low η: Water, Air.
Temperature Effect: Heating a liquid (like oil) makes it thinner (Viscosity decreases). Heating a gas makes it thicker (Viscosity increases).
8.1 Stokes’ Law and Terminal Velocity
Imagine dropping a small steel ball into a jar of glycerine.
1. Initially, gravity pulls it down, and it accelerates.
2. But as it speeds up, the fluid friction (Viscous Drag) increases.
3. Eventually, the upward Drag + Buoyancy exactly balances the downward Weight.
4. The Net Force becomes Zero. The ball stops accelerating and falls at a constant speed.
This constant speed is called Terminal Velocity.
Real Life: Raindrops fall from clouds high in the sky. If there were no air viscosity, they would hit us like bullets! Thankfully, air resistance slows them down to a safe terminal velocity.
9. Surface Tension: The Stretched Skin
Have you noticed that water drops are always spherical? Or that a steel needle can float on water if placed gently?
This happens because the surface of a liquid behaves like a stretched elastic membrane. This property is called Surface Tension.
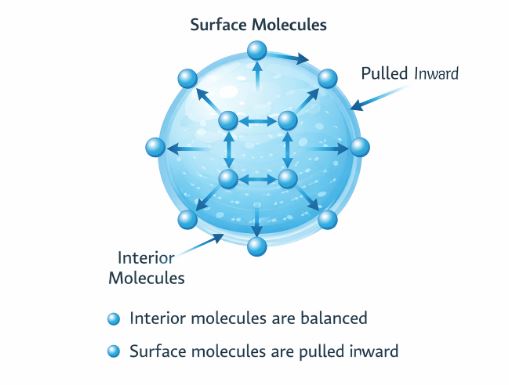
Figure 3: Molecular view. Surface molecules are pulled inward, creating tension.
9.1 Why does it happen? (Cohesive Forces)
Molecules of water attract each other (Cohesion).
– A molecule deep inside is pulled equally in all directions. Net force = 0.
– A molecule on the surface has water only below it. So, it gets pulled Downwards.
This inward pull tries to shrink the surface area to the minimum possible.
For a given volume, the shape with the minimum surface area is a Sphere. That’s why raindrops are round!
9.2 Surface Energy
To increase the surface area (like blowing a soap bubble), you have to pull molecules up against this inward force. You have to do Work. This work gets stored as Surface Energy.
Work Done = Surface Tension (S) × Change in Area (ΔA)
9.3 Capillarity (Water climbing up)
If you dip a very thin glass tube (capillary) in water, the water rises up in the tube against gravity!
Why? Because water loves glass (Adhesion > Cohesion). The water climbs the walls, and surface tension pulls the liquid column up.
This is how plants transport water from roots to leaves, and how a towel soaks up moisture.
9.4 Detergents and Surface Tension
Why can’t we wash oily clothes with just water?
Water has high surface tension; it tends to form drops and doesn’t spread into the tiny pores of the cloth. Oil is hydrophobic (hates water).
Soap/Detergent reduces the surface tension of water. This helps water spread and penetrate the cloth fibers. It also grabs the oil molecules and washes them away.
10. Practice Questions & Detailed Solutions
Let’s verify your understanding with some conceptual and numerical problems.
Part A: Multiple Choice Questions (MCQ)
- Water flows from a pipe of diameter 4 cm into a pipe of diameter 2 cm. The speed of the water in the second pipe is:
(a) 4 times speed in first pipe.
(b) 2 times speed in first pipe.
(c) Half speed in first pipe.
(d) 1/4th speed in first pipe.Solution: (a).
Reasoning: From Equation of Continuity,
A₁v₁ = A₂v₂.
AreaA = π(d/2)² ∝ d².
If diameter halves (4 to 2), the Area becomes (1/2)² = 1/4th.
To keep flow constant, Velocity must become 4 times. - Why does a shower curtain often get pulled inwards when the water is turned on?
(a) Pascal’s Law (b) Archimedes’ Principle (c) Bernoulli’s Principle (d) Surface TensionSolution: (c).
Reasoning: The flowing water drags air with it, creating a high-speed air region inside the shower. According to Bernoulli, High Speed = Low Pressure. The air outside is still (High Pressure). The outside pressure pushes the curtain inwards.
- An object is falling through the air at its terminal velocity. The net force on the object is:
(a) Downwards (Weight). (b) Upwards (Drag). (c) Zero. (d) Less than weight.Solution: (c).
Reasoning: Terminal velocity means constant velocity. Constant velocity means acceleration is Zero. By Newton’s Second Law (F=ma), if a=0, then Net Force = 0. The forces (Gravity down vs Drag+Buoyancy up) are perfectly balanced.
Part B: Short Answer Questions
- Q: Why is it easier to cut something with a sharp knife than a dull one?
Answer: This relies on the concept of Pressure.
Formula:Pressure = Force / Area.
A sharp knife has an extremely thin edge, meaning the Area (A) of contact is very small. For the same force applied by your hand, the small Area results in massive Pressure. This high pressure easily breaks the molecular bonds of the vegetable. A dull knife has a larger area, reducing the pressure. - Q: Why do air bubbles in a fish tank get larger as they rise to the surface?
Answer:
1. At the bottom of the tank, the pressure is high (Atmospheric + Water Pressure ρgh).
2. As the bubble rises, the depth ‘h’ decreases, so the water pressure decreases.
3. According to Boyle’s Law (for gases), Pressure is inversely proportional to Volume (PV = constant).
4. Since the external pressure decreases, the volume of the gas inside the bubble expands, making it larger.
Part C: Long Answer Questions (Numerical Solving)
- Q: A large, open water tank is 20 m tall and filled to the top. A small hole is punched in the side 4 m above the ground. What is the speed of the water as it exits the hole? (Take g = 9.8 m/s²)
Answer:
This problem uses Torricelli’s Law (a special case of Bernoulli’s principle).
The speed of efflux depends on the depth of the hole below the free surface of the water.
Given:
Total height of tank = 20 m.
Height of hole from ground = 4 m.
Depth of hole from surface (h) = Total – Height from ground = 20 – 4 = 16 m.Formula:
v = √(2gh)
v = √(2 × 9.8 × 16)
v = √(313.6)
v ≈ 17.7 m/s.
Note: Many students mistake ‘h’ for height from the ground. Always measure depth from the top surface!
Read Also:
Class-11 Chapter 8- Mechanical Properties of Solids
For more check official website of
NCERT
