Start Chapter MCQ Quiz
Detailed Notes: Class 11 Physics Chapter 8 Mechanical Properties of Solids notes
1. Introduction: The Reality of “Solid” Objects | Class 11 Physics Chapter 8 Mechanical Properties of Solids notes
Hello students! Welcome to a new unit. Up until now, in our study of mechanics (like rotation and motion), we assumed something very convenient. We assumed that objects were Rigid Bodies. We pretended that when you hit a cricket ball or rotate a wheel, its shape never changes. The distance between any two atoms inside it stays fixed forever.
But looking around the real world, we know this is a lie. Nothing is perfectly rigid.
If you sit on a sofa, the cushion sinks. If you stretch a rubber band, it elongates. Even a steel bridge bends slightly when a heavy truck drives over it. Even the hardest diamond can be deformed if you apply enough force.
In this chapter, we are going to study the Mechanical Properties of Solids. We will explore why things change shape, how they return to their original shape, and when they break. This science is the foundation of Civil Engineering and Mechanical Engineering. Without it, every building we build would collapse!
1.1 Elasticity and Plasticity
Imagine you have a piece of rubber and a piece of chewing gum. You stretch both of them.
- The Rubber: When you let go, it snaps back to its original length.
- The Chewing Gum: When you let go, it stays stretched and messy.
This difference is the core of this chapter.
1. Elasticity:
This is the “memory” of a material. It is the property by virtue of which a body tends to regain its original size and shape after the applied force is removed.
Examples: Steel, Rubber, Quartz, Human Skin.
2. Plasticity:
This is the “forgetfulness” of a material. It is the property by which a body does not regain its original shape and gets permanently deformed.
Examples: Putty, Mud, Chewing Gum, Paraffin Wax.
1.2 The Microscopic View: Atoms as Springs
Why do solids behave like this? Let’s zoom in to the atomic level.
Solids are made of atoms arranged in a lattice. Imagine these atoms are small balls connected to their neighbors by invisible Springs.
– When you pull the object, you are stretching these inter-atomic springs. They want to pull back (Restoring Force).
– When you squash the object, you are compressing the springs. They want to push back.
This internal “springiness” of chemical bonds is the origin of Elasticity.
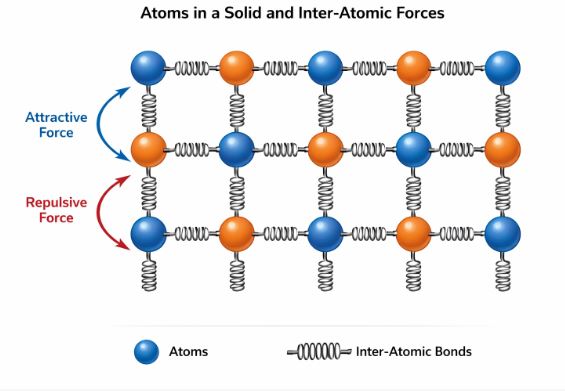
Figure 1: The Spring-Ball Model of Solids. Stretching the solid stretches the bonds between atoms.
2. Stress: The Internal Fighter
When you pull a rubber band, you apply an External Deforming Force.
But the rubber band doesn’t just give up. It fights back! Inside the rubber, millions of molecular bonds are pulling back, trying to restore the original shape.
Definition: Stress is the Internal Restoring Force developed per unit area of the body.
In equilibrium, the internal restoring force is equal in magnitude to the external applied force. So, for calculation purposes, we use the applied force.
Formula: Stress (σ) = Force (F) / Area (A)
SI Unit: N/m² or Pascal (Pa).
Dimensional Formula: [ML⁻¹T⁻²] (Same as Pressure).
2.1 Types of Stress
Depending on how the force is applied, stress comes in three flavors:
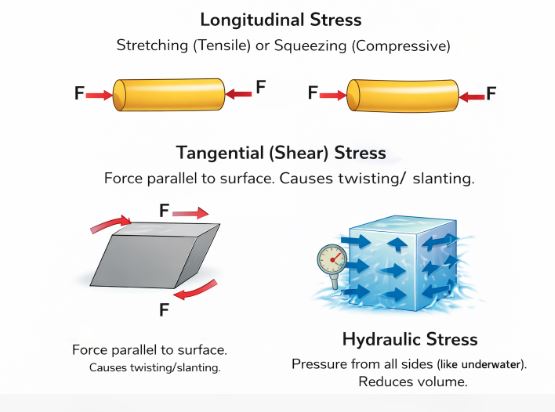
Figure 2: The three ways to stress a body.
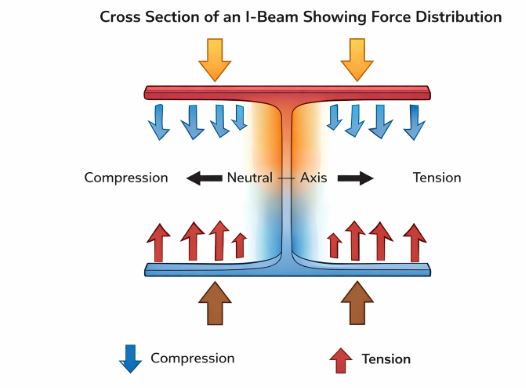
A. Longitudinal Stress
This happens when the force acts along the length of a cylinder or wire.
– Tensile Stress: When the force pulls the object to elongate it.
– Compressive Stress: When the force pushes the object to shorten it.
B. Tangential (Shearing) Stress
Imagine a thick book on a table. If you push the top cover sideways while keeping the bottom fixed, the book slants. The pages slide over each other.
Here, the force is applied Parallel to the surface area.
C. Hydraulic Stress
This happens to a ball thrown deep into the ocean. The water pressure pushes it from every single direction perpendicular to the surface. The ball shrinks in volume but keeps its shape.
3. Strain: The Measure of Deformation
Stress is the cause; Strain is the effect.
When you stress an object, it changes dimensions. Strain tells us “how much” it changed relative to its original size.
Formula: Strain (ε) = Change in Dimension / Original Dimension
Unit: Strain is a ratio of two similar quantities (like length/length). Therefore, it has NO UNIT and NO DIMENSIONS.
3.1 Types of Strain
Just like stress, strain has three corresponding types:
- Longitudinal Strain: Change in Length per unit Original Length.
ε_l = ΔL / L - Shearing Strain: The angle by which a vertical face gets tilted.
ε_s = tan θ ≈ θ = Δx / L(Relative displacement / Length). - Volume Strain: Change in Volume per unit Original Volume.
ε_v = ΔV / V
4. Hooke’s Law
Robert Hooke, a contemporary of Newton, did many experiments with springs and wires. He found a very simple relationship.
Statement: “For small deformations, Stress is directly proportional to Strain.”
Stress ∝ Strain
Stress = k × Strain
This constant of proportionality ‘k’ is called the Modulus of Elasticity.
This law is valid only up to a certain limit called the Elastic Limit.
5. The Stress-Strain Curve: A Material’s Biography
If we take a metal wire and stretch it more and more while measuring the stress and strain, we get a graph. This graph tells us the complete life story of that material—from being elastic to breaking apart.
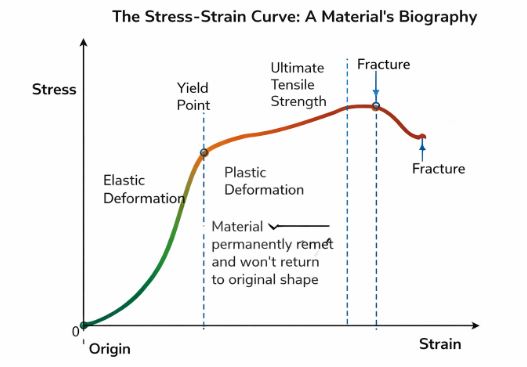
Figure 3: The Stress-Strain Curve for a ductile metal.