1. Introduction: The Magic of Change (Chemical Reactions) | Chapter-1 Chemical Reactions & Equations
Hello Students! Welcome to the world of Chemistry. Chemistry is basically the study of change. Look around you—everything is changing. Milk turns into curd, iron nails get rusty, and food gets cooked.
Let’s start with a simple example from your daily life. Think about baking a cake. When you are in the kitchen, you start with separate ingredients: white flour, wet eggs, and sweet sugar. These are your starting materials. You mix them into a batter and put them in the oven (add heat). What comes out? A fluffy, delicious cake!
Now, ask yourself: Can you get the eggs back from the cake? Can you separate the sugar from the baked bread? No! The change is permanent. The properties of the cake are completely different from the flour and eggs. This creates a new substance. In science, we call this permanent transformation a Chemical Reaction.
Reactants and Products
Every chemical reaction has two main parts:
- Reactants: These are the substances that take part in the reaction. In our cake example, the flour, eggs, and sugar are the reactants. They are the “Ingredients.”
- Products: These are the new substances formed after the reaction. The cake is the product. It is the “Result.”
How do we know a reaction has happened?
Sometimes, changes happen silently, but often, chemistry gives us clues! You can tell a chemical reaction has taken place if you observe any of these four major changes:
[Image of chemical reaction indicators]
- Change in Color: Have you seen an old iron gate turn reddish-brown? That’s rust. The iron (grey) reacted with air and moisture to form rust (brown).
- Change in State: Imagine burning a solid candle wax. As it burns, it turns into smoke (gas) and water vapor. A solid turned into a gas!.
- Evolution of Gas: If you drop an antacid tablet into water, you see fizzy bubbles coming out instantly. This gas production proves a reaction is happening.
- Change in Temperature: Have you ever held a handful of detergent powder and added a little water? Your palm feels warm. This is because the reaction releases heat.
2. Chemical Equations: The Language of Chemistry
Imagine if you had to write every reaction in full sentences: “One atom of Zinc reacts with one molecule of Sulphuric Acid to produce Zinc Sulphate and Hydrogen.” That is too long and boring!
Chemists are smart; they use a shorthand method called Chemical Equations. It’s like sending a text message using emojis and short forms instead of writing a whole letter.
Word Equation vs. Formula Equation
- Word Equation: Hydrogen + Oxygen → Water. (Simple, but still long).
- Chemical Formula Equation:
H₂ + O₂ → H₂O. (Short, precise, and scientific).
If you look closely at the equation H₂ + O₂ → H₂O, you will see a problem. On the left, there are 2 Oxygens. On the right, there is only 1 Oxygen. Where did the other Oxygen go? This is called a Skeletal Equation (or an unbalanced equation).
Why Must We Balance Equations?
Nature follows strict rules. The most important one here is the Law of Conservation of Mass. This law states that matter cannot be created or destroyed.
This means you cannot lose an atom during a reaction, and you cannot magically create a new one. The number of atoms of each element on the Reactant side (Left) MUST be equal to the number of atoms on the Product side (Right).
Making Equations More Informative:
To make our equation tell the full story, we add State Symbols next to the formulas:
(s)= Solid (like ice or metal)(l)= Liquid (like water)(g)= Gas (like steam or oxygen)(aq)= Aqueous (This means “dissolved in water”)
3. Types of Chemical Reactions
Just like people interact in different ways (making friends, breaking up, fighting, swapping groups), chemicals also react in specific patterns. We can group them into four main types.
1. Combination Reaction (The Friendship)
Imagine two separate dancers coming together to perform as a couple. In chemistry, when two or more simple reactants combine to form one single product, it is a Combination Reaction.
General Formula: A + B → AB
Example: Burning of Coal. Carbon (C) and Oxygen (O₂) combine to form Carbon Dioxide (CO₂).
2. Decomposition Reaction (The Breakup)
This is the exact opposite of combination. Here, a single large reactant breaks down into two or simpler products. Think of a complex lego tower breaking into individual bricks.
General Formula: AB → A + B
Important Note: Breaking things requires energy! You can’t just stare at the chemical and expect it to break. We need to supply energy in three forms:
- Heat (Thermal Decomposition): Example: Heating Lead Nitrate.
- Light (Photolytic Decomposition): Example: Silver Chloride turning grey in sunlight.
- Electricity (Electrolytic Decomposition): Example: Breaking water into H₂ and O₂ using electricity.
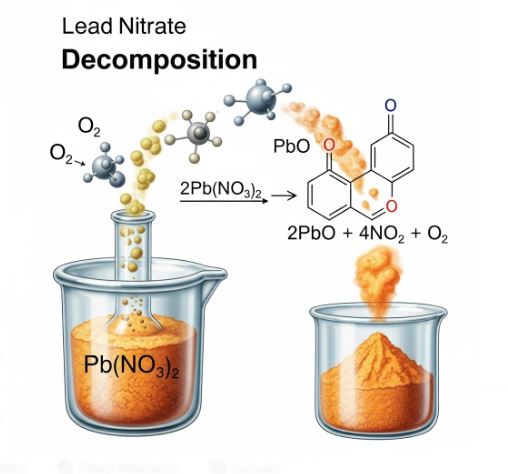
Visual Evidence: When we heat white Lead Nitrate powder, we see reddish-brown fumes. These fumes are Nitrogen Dioxide (NO₂). This confirms the decomposition.
3. Displacement Reaction (The Bully)
Imagine a strong player replacing a weaker player in a team. In chemistry, a more reactive element kicks out (displaces) a less reactive element from its solution.
General Formula: A + BC → AC + B
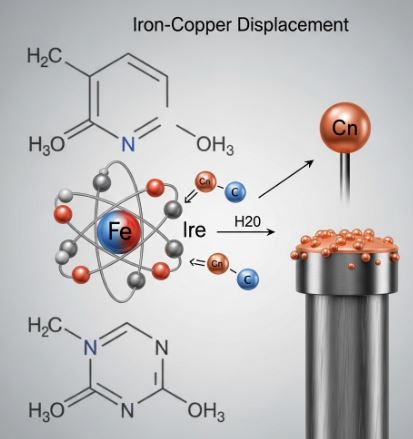
The Iron Nail Experiment: If you dip a clean Iron nail into a blue Copper Sulfate solution, magic happens! After some time, the blue color fades to green (Iron Sulfate formed), and the Iron nail gets a brownish coating. Why? Because Iron is stronger (more reactive) than Copper and kicked it out!
4. Double Displacement Reaction (The Partner Swap)
This reaction is like two couples swapping dance partners. Two different compounds react by exchanging their ions to form two new compounds.
General Formula: AB + CD → AD + CB
Usually, one of the new products formed is an insoluble solid that settles at the bottom. This solid is called a Precipitate.

Example: When you mix Potassium Iodide and Lead Nitrate (both clear liquids), you suddenly get a bright Yellow Precipitate of Lead Iodide.
4. Oxidation and Reduction (Redox Reactions)
In many reactions, oxygen or hydrogen is exchanged. We call these Redox reactions because Reduction and Oxidation happen at the same time.
[Image of oxidation and reduction cycle]
Let’s simplify definitions:
- Oxidation: When a substance Gains Oxygen OR Loses Hydrogen. (Think: “Oxidation is adding Oxygen”).
- Reduction: When a substance Loses Oxygen OR Gains Hydrogen. (Think: “Reduction is reducing Oxygen”).
Redox in Everyday Life
Chemical reactions aren’t just in the lab; they are destroying bridges and spoiling food!
- Corrosion: Have you noticed copper statues turning green or silver jewelry turning black? This is corrosion. It is the slow eating away of metals by moisture and acids in the air. Rusting of iron is the most common form of corrosion.
- Rancidity: Have you ever tasted chips from an open packet left for days? They taste “stale” and smell bad. This is Rancidity. The fats and oils in the food have oxidized (reacted with oxygen).
Fun Fact: That is why chips packets are so puffy! They aren’t filled with air; they are flushed with Nitrogen gas. Nitrogen is unreactive and prevents oxygen from touching the chips, keeping them fresh.
5. Extensive Practice Set (With Teacher’s Solutions)
Now that we have learned the concepts of Chapter-1 Chemical Reactions & Equations , let’s solve some important questions. I have written the solutions just like how you should write them in your exam.
Part A: Multiple Choice Questions (MCQ)
- A student mixes two clear liquids and the beaker becomes very cold. What type of reaction is this?
(a) Combination (b) Exothermic (c) Endothermic (d) DecompositionSolution: (c) Endothermic.
Reasoning: “Endo” means “in” and “Thermic” means “heat”. Reactions that absorb heat from the surroundings make the container feel cold. - The equation Mg(s) + O₂(g) → MgO(s) is an example of:
(a) Combination only (b) Oxidation and Combination (c) Decomposition and Oxidation (d) Double DisplacementSolution: (b) Oxidation and Combination.
Reasoning: It is a Combination because two things (Mg + O) became one (MgO). It is ALSO Oxidation because Magnesium gained Oxygen. - What is the primary purpose of balancing a chemical equation?
(a) To make it look simpler. (b) To satisfy the Law of Conservation of Mass. (c) To indicate temperature. (d) To show reactivity.Solution: (b) To satisfy the Law of Conservation of Mass.
Reasoning: We cannot create or destroy atoms, so we must balance them. - 2AgNO₃(aq) + Zn(s) → Zn(NO₃)₂(aq) + 2Ag(s). This is an example of:
(a) Decomposition (b) Double Displacement (c) Combination (d) DisplacementSolution: (d) Displacement.
Reasoning: Look closely—Zinc (Zn) is sitting alone, and then it kicks Silver (Ag) out of the Nitrate compound. A single element replacing another is Displacement. - When food becomes rancid, what has reacted with the fats and oils?
(a) Nitrogen (b) Water (c) Oxygen (d) Carbon DioxideSolution: (c) Oxygen.
Reasoning: Rancidity is basically the oxidation of food. Oxygen is the culprit here.
Part B: Short Answer Questions
- A shiny silver anklet turns black after several months. Name this process and the type of chemical change.
Solution:
Process Name: Corrosion (specifically referred to as the tarnishing of silver).
Explanation: This is a chemical change where silver metal reacts with sulfur compounds present in the air.
Product: It forms a black coating of Silver Sulfide (Ag₂S). - Translate into a balanced chemical equation: “Nitrogen gas reacts with hydrogen gas at high pressure to form ammonia gas.”
Solution:
Step 1 (Skeletal): N₂ + H₂ → NH₃
Step 2 (Balancing): We have 2 Nitrogens on the left, so we need 2 NH₃ on the right. Now we have 6 Hydrogens on the right (2×3), so we need 3 H₂ on the left.
Final Equation: N₂(g) + 3H₂(g) → 2NH₃(g). - Why is photosynthesis considered an endothermic reaction?
Solution:
An endothermic reaction is one that absorbs energy. During photosynthesis, green plants absorb solar energy (sunlight) to break down carbon dioxide and water to form glucose. Since energy is taken IN, it is endothermic. - Balance the equation: C₃H₈(g) + O₂(g) → CO₂(g) + H₂O(g)
Solution:
Let’s balance it step-by-step:
1. Carbon: 3 on left (C₃), so put 3 in front of CO₂. (C₃H₈ + O₂ → 3CO₂ + H₂O)
2. Hydrogen: 8 on left (H₈), so put 4 in front of H₂O. (C₃H₈ + O₂ → 3CO₂ + 4H₂O)
3. Oxygen: Right side now has (3×2) + (4×1) = 10 Oxygens. So we put 5 in front of O₂.
Final: C₃H₈(g) + 5O₂(g) → 3CO₂(g) + 4H₂O(g).
Part C: Long Answer Questions
- Differentiate between displacement and double displacement reactions. Provide a unique, balanced chemical equation for each.
Solution:
1. Displacement Reaction:
Definition: A reaction in which a more reactive element displaces a less reactive element from its compound.
Pattern: A + BC → AC + B
Example: Zinc displaces Copper from Copper Sulfate.
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)2. Double Displacement Reaction:
Definition: A reaction in which there is an exchange of ions between the reactants to form new compounds.
Pattern: AB + CD → AD + CB
Example: Silver Nitrate reacts with Sodium Chloride.
AgNO₃(aq) + NaCl(aq) → AgCl(s) + NaNO₃(aq) - Explain oxidation and reduction using the reaction: MnO₂(s) + 4HCl(aq) → MnCl₂(aq) + 2H₂O(l) + Cl₂(g). Identify the substance oxidized and reduced.
Solution:
To find what is oxidized and reduced, we track the Oxygen and Hydrogen:
1. Substance Reduced: Look at MnO₂. It starts with Oxygen but becomes MnCl₂ (no oxygen). Removal of Oxygen is Reduction. So, MnO₂ is reduced.
2. Substance Oxidized: Look at HCl. It starts with Hydrogen but becomes Cl₂ (no hydrogen). Removal of Hydrogen is Oxidation. So, HCl is oxidized.
Read Also:
Class 9 Science Notes
Chapter-1 Chemical Reactions & Equations
For more check official website of
NCERT

