
Start Chapter MCQ Quiz
Comprehensive Guide: Class 12 Physics Chapter 11 Dual Nature of Radiation and matter Notes
Introduction | Class 12 Physics Chapter 11 Dual Nature of Radiation and matter Notes
Hello students! Welcome to one of the most fascinating turning points in modern physics. Up until now, we have been very comfortable with a specific idea: Light is a wave. The famous Maxwell’s equations and Heinrich Hertz’s experiments beautifully proved the electromagnetic wave nature of light. We even saw this wave nature in action when we studied interference and diffraction.
But physics is full of surprises. Towards the end of the 19th century, scientists started playing with discharge tubes and discovered electrons. They also noticed that when certain types of light hit a metal, something strange happened—electrons popped out. When they tried to explain this using our trusty wave theory, it completely failed! This chapter is all about that crisis, how Albert Einstein solved it by proposing that light behaves like particles, and how Louis de Broglie later blew our minds by suggesting that matter can behave like waves. Let us dive into this “Dual Nature” of our universe.
1. Electron Emission and The Work Function
1.1 What Keeps Electrons Inside a Metal?
We know that metals are excellent conductors because they are full of free electrons. However, the word “free” is a bit misleading. They are free to roam inside the metal, but they cannot easily leave the surface. Imagine a marble rolling around the bottom of a bowl. It can move freely at the bottom, but it needs a strong push to jump out.
Similarly, the moment an electron tries to escape, the metal surface gets a positive charge and violently yanks the electron back inside. The electron is trapped by these attractive ionic forces. To break free, the electron needs a specific minimum amount of energy.
1.2 The Work Function ($\phi_0$)
This brings us to a crucial definition for your Board Exams: Work Function. It is the absolute minimum energy required by an electron to just escape from the metal surface.
- Symbol: It is denoted by $\phi_0$.
- Unit: Instead of Joules, we usually measure it in electron volts (eV) because dealing with atomic energies in Joules gives us unnecessarily tiny numbers. One electron volt is the energy gained by an electron accelerated through a 1-volt potential difference ($1\text{ eV}=1.602\times10^{-19}\text{ J}$).
- Dependence: The work function is not a universal constant. It changes depending on the nature of the metal and the condition of its surface.
Teacher’s Note: Remember that free electrons inside the metal have different kinetic energies based on their distribution. The work function is the energy needed by the most energetic, surface-level electron to escape. Deeper electrons would need even more energy!.
1.3 Types of Electron Emission
How can we give these electrons the energy they need to overcome the work function? We have three main methods:
- Thermionic Emission: We simply heat the metal. The thermal energy gets transferred to the electrons, giving them enough kinetic energy to boil off the surface.
- Field Emission: We apply a massively strong electric field (around $10^8\text{ V/m}$) to literally rip the electrons out of the metal. This happens in spark plugs.
- Photoelectric Emission: We shine light of a suitable, high frequency onto the metal surface, causing electrons to be ejected. These ejected electrons are called photoelectrons. This is the star of our chapter!
2. The Photoelectric Effect: Historical Observations
The photoelectric effect wasn’t discovered in a single day. It was a puzzle pieced together by brilliant minds.
Heinrich Hertz (1887): While trying to prove the existence of electromagnetic waves, Hertz noticed something odd. When he illuminated his metal emitter plates with ultraviolet (UV) light, the high-voltage sparks in his detector loop became much stronger. The UV light was somehow helping charges escape the metal.
Wilhelm Hallwachs and Philipp Lenard (1886-1902): They took Hertz’s observation further. Lenard used an evacuated glass tube with two metal plates. He found that when UV light hit the emitter plate, a current flowed in the circuit. When the light stopped, the current vanished instantly. The light was ejecting negatively charged particles (electrons) that traveled to the positive collector plate.
Hallwachs did a brilliant secondary experiment. He took a negatively charged zinc plate and shined UV light on it. The plate lost its negative charge!. When he shined UV light on a neutral zinc plate, it became positively charged. This proved beyond a doubt that the light was knocking negative charges out of the metal.
The Threshold Frequency Discovery: They also noticed a vital rule. If the frequency of the light was too low, absolutely nothing happened. No electrons were emitted, no matter how bright the light was. This critical minimum frequency is called the Threshold Frequency, and it depends purely on the material of the metal plate. Zinc needs high-frequency UV light, but alkali metals like sodium and potassium are so sensitive that even visible light can knock their electrons loose.
3. Experimental Study of the Photoelectric Effect
Pay close attention here, students. The graphs in this section are highly tested in the CBSE board exams. You must understand how to draw and interpret them.
The Setup: The experiment uses an evacuated glass tube containing a photosensitive emitter plate (C) and a collector plate (A). Light passes through a quartz window and hits plate C. Emitted electrons travel to plate A, creating a photocurrent measured by a microammeter. A battery is used to change the potential difference between the plates, and a commutator can reverse the polarity (making plate A positive or negative).
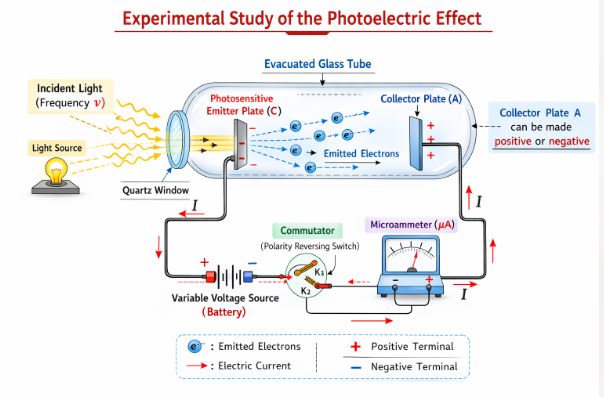
Figure-1: Schematic of the experimental setup to study the photoelectric effect. Light hits the photosensitive plate C, ejecting electrons that travel to collector plate A.
3.1 Effect of Intensity of Light on Photocurrent
Let’s keep the frequency of the light and the voltage constant. What happens if we just make the light brighter (increase its intensity)?.
The result is a straight line passing through the origin. The photocurrent increases linearly with the intensity of incident light.
Why? Because higher intensity means more light energy hitting the plate, which knocks out a proportionally higher number of photoelectrons per second.
3.2 Effect of Potential on Photocurrent
Now, let’s keep the light intensity ($I_1$) and frequency fixed. We start making the collector plate (A) more and more positive.
The positive plate attracts the electrons strongly, so the photocurrent rises. Eventually, it maxes out. All the electrons being emitted are safely reaching the collector. This maximum flat line is called the Saturation Current.
But what if we reverse the battery? We make plate A negative. Now, plate A repels the incoming electrons. The slow electrons get pushed back, and the photocurrent drops. Only the fastest, most energetic electrons can fight their way to plate A.
If we make plate A negative enough, even the absolute fastest electron is stopped, and the photocurrent becomes exactly zero.
This specific negative voltage is called the Stopping Potential ($V_0$) or cut-off potential. It gives us a direct measurement of the maximum kinetic energy ($K_{max}$) of the electrons:
$$K_{max} = e V_0$$
Crucial Observation: If we repeat this with brighter light ($I_3 > I_2 > I_1$), the saturation current is higher (more electrons), BUT the stopping potential remains exactly the same!. Conclusion: Maximum kinetic energy depends on the emitter material and light source, but is entirely independent of light intensity!.
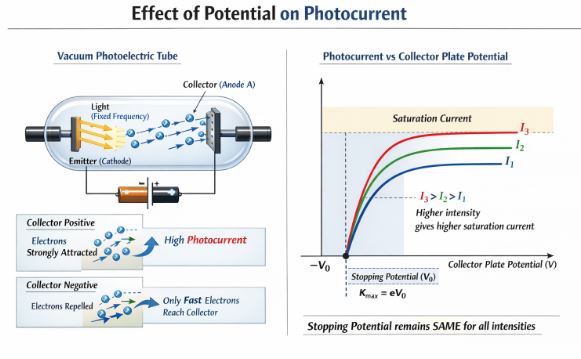
Figure-2: The stopping potential remains constant even when the intensity of light is increased. Only the saturation current changes.
3.3 Effect of Frequency on Stopping Potential
Finally, let’s keep the intensity constant but change the color (frequency, $\nu$) of the light.
We find that higher frequencies require a more negative stopping potential to halt the electrons. For frequencies $\nu_3 > \nu_2 > \nu_1$, the stopping potentials follow $V_{03} > V_{02} > V_{01}$.
This means higher frequency light ejects faster electrons with greater kinetic energy. If we plot Stopping Potential vs. Frequency, we get a beautiful straight line. This graph shows two things clearly:
- Stopping potential varies linearly with frequency.
- There is an x-intercept! Below a certain threshold frequency ($\nu_0$), the stopping potential is zero, meaning no emission occurs at all.
Lastly, the emission process is virtually instantaneous. Even if the light is extremely dim, if the frequency is right, electrons pop out in less than $10^{-9}$ seconds!.
4. The Crisis: Failure of the Wave Theory
Now, put on your classical physics hats. According to the wave theory of light, energy is spread out continuously over an entire wavefront. Let’s see why this theory miserably fails to explain the photoelectric effect:
- The Intensity Problem: Wave theory says a brighter, more intense wave has larger electric and magnetic amplitudes. So, a brighter wave should impart more energy to each electron. The kinetic energy of the electrons should increase with intensity. But our experiments just proved that kinetic energy is completely independent of intensity!.
- The Threshold Frequency Problem: According to waves, frequency shouldn’t matter for emission. If you shine a low-frequency red light on a metal for a long enough time, the continuous wave energy should eventually add up, giving the electron enough energy to escape. Therefore, a threshold frequency should not exist. But reality shows us that without reaching the threshold frequency, no electrons ever emit, even if you wait a century.
- The Time Lag Problem: A wave spreads its energy over millions of electrons on the surface. Calculations show it would take hours for a single electron to gather enough energy from a dim wave to overcome the work function. But experiments show emission is instantaneous ($< 10^{-9}$ s).
The wave picture was broken. Physics needed a new hero.
5. Einstein’s Photoelectric Equation: The Photon Arrives
In 1905, Albert Einstein proposed a radical, mind-bending idea. He said we must stop thinking of light as a continuous wave in this scenario. Instead, light energy travels in tiny, discrete packets or bundles. He called these quanta of energy, which we now know as Photons.
The energy of a single photon is strictly determined by its frequency:
$$E = h\nu$$
Where $h$ is Planck’s constant and $\nu$ is the frequency.
Einstein pictured the photoelectric effect not as a wave washing over a beach, but as a game of billiards. One photon hits exactly one electron. It transfers its entire energy ($h\nu$) to that single electron instantly.
The electron uses a part of this energy to pay the “exit toll” (the work function, $\phi_0$) to escape the metal. Whatever energy is left over becomes the electron’s maximum kinetic energy. This gives us Einstein’s famous Photoelectric Equation:
$$K_{max} = h\nu – \phi_0$$
How Einstein’s Equation Solves the Mysteries:
- Intensity Explained: In Einstein’s view, increasing intensity just means shooting more photons per second. More photons mean more collisions, which means more electrons emitted (higher photocurrent). But the energy of each photon ($h\nu$) hasn’t changed. Thus, the maximum kinetic energy of the electrons remains exactly the same, independent of intensity.
- Threshold Frequency Explained: For an electron to escape, the incoming photon’s energy must be at least equal to the work function ($h\nu \ge \phi_0$). If we solve for the minimum frequency $\nu_0$, we get:
$$\nu_0 = \frac{\phi_0}{h}$$
. If the incoming light frequency is less than $\nu_0$, the photon simply doesn’t have enough energy to free the electron. No emission is possible. - Instantaneous Emission Explained: Because it’s a one-on-one particle collision, the energy transfer is immediate. There is no “gathering” of wave energy over time.
Teacher’s Note for Derivations: We know that $K_{max} = eV_0$. Substituting this into Einstein’s equation gives:
$$eV_0 = h\nu – \phi_0$$
$$V_0 = \left(\frac{h}{e}\right)\nu – \frac{\phi_0}{e}$$
.
Look closely! This is the equation of a straight line ($y = mx + c$). The slope of the Stopping Potential vs. Frequency graph is absolutely constant for all materials and equals $\frac{h}{e}$. Robert Millikan later verified this equation experimentally with incredible precision, proving Einstein right and accurately calculating Planck’s constant.
6. The Particle Nature of Light: The Photon
Thanks to the photoelectric effect, and later Arthur Compton’s X-ray scattering experiments in 1924, the particle nature of light was firmly established. Here is the definitive summary of the photon picture:
- When interacting with matter, radiation behaves as a stream of particles called photons.
- Every photon has energy $E = h\nu = \frac{hc}{\lambda}$ and momentum $p = \frac{h\nu}{c} = \frac{h}{\lambda}$. They travel at the speed of light, $c$.
- All photons of a specific wavelength or frequency have the exact same energy and momentum, regardless of the light’s intensity. Increasing intensity only increases the number of photons crossing an area, not their individual energy.
- Photons are electrically neutral. They do not get deflected by electric or magnetic fields.
- In a photon-electron collision, total energy and total momentum are conserved. However, the photon itself might be entirely absorbed (its number is not conserved).
7. The Wave Nature of Matter (de Broglie Hypothesis)
If light—which we thought was a pure wave—can act like a particle, could particles of matter act like waves?.
In 1924, a bold French physicist named Louis Victor de Broglie proposed exactly this. He argued that nature is symmetrical. Since energy exhibits a dual nature, matter must also possess a dual (wave-particle) character.
De Broglie suggested that every moving particle has an associated wave, called a matter wave or de Broglie wave. The wavelength ($\lambda$) of this matter wave is perfectly linked to the particle’s momentum ($p$):
$$\lambda = \frac{h}{p} = \frac{h}{mv}$$
.

Figure-3: The dualism of matter. The equation $\lambda = h/p$ bridges the wave aspect ($\lambda$) with the particle aspect (momentum, p).
Why don’t we see everyday objects acting like waves?
You might wonder: “If I throw a cricket ball, why doesn’t it diffract and wave around?” Let’s look at the math. The de Broglie wavelength is inversely proportional to mass ($m$). Planck’s constant ($h$) is incredibly tiny ($6.63 \times 10^{-34}\text{ J s}$).
If a 0.12 kg ball moves at 20 m/s, its wavelength is:
$$\lambda = \frac{6.63 \times 10^{-34}}{0.12 \times 20} = 2.76 \times 10^{-34} \text{ m}$$
. This wavelength is so unfathomably small that it is impossible to measure. That is why macroscopic objects don’t show wave properties.
But for an electron, with its extraordinarily tiny mass ($9.11 \times 10^{-31}\text{ kg}$), moving at high speeds, the wavelength comes out to around $0.135 \text{ nm}$. This is perfectly measurable and is comparable to X-ray wavelengths!. In the sub-atomic domain, the wave nature of matter rules.
8. Real-Life Applications & Examples
- Solar Panels: Though they rely on the internal photovoltaic effect, the core principle is the same. Light photons hit the semiconductor material, providing energy to knock electrons loose and create a useful electric current.
- Light Meters in Cameras: These devices use photosensitive materials that measure the intensity of light by evaluating the photoelectric current produced, helping photographers get the perfect exposure.
- Electron Microscopes: Leveraging de Broglie’s wave nature of matter, electron microscopes use the tiny wavelengths of accelerated electrons to achieve magnifications millions of times greater than light microscopes, allowing us to see individual atoms!
9. Common Student Misconceptions (Teacher’s Desk)
Misconception 1: “A brighter light will give electrons more kinetic energy.”
Correction: False. A brighter light (higher intensity) only increases the number of photons, not their individual energy. Kinetic energy is strictly determined by the light’s frequency (color).
Misconception 2: “All electrons emitted in the photoelectric effect have the same kinetic energy.”
Correction: No. The equation $K_{max} = h\nu – \phi_0$ only gives the maximum possible kinetic energy. Electrons deeper inside the metal lose energy colliding on their way out and emerge with less kinetic energy.
Misconception 3: “Light is definitively a particle now.”
Correction: Light is dual. It exhibits wave properties (interference, diffraction) in propagation, and particle properties (photoelectric effect, Compton scattering) in interaction with matter. The experiment decides which nature is observed.
10. CBSE Practice Set with Solutions
Very Short Answer Type (1-2 Marks)
Q1: Define Work Function of a metal. On what factors does it depend?
Ans: Work function is the minimum amount of energy required by an electron to just escape from the metal surface. It is denoted by $\phi_0$ and depends on the nature of the metal and its surface properties.
Q2: Why does photoelectric emission fail to occur if the frequency of incident light is less than the threshold frequency, regardless of the light’s intensity?
Ans: According to Einstein, a single electron absorbs a single photon. If the photon’s frequency $\nu$ is less than the threshold $\nu_0$, the photon’s energy ($h\nu$) is strictly less than the work function ($\phi_0$). Therefore, the electron can never gain enough energy to escape, even if millions of such low-energy photons strike the surface (high intensity).
Short Answer Type (3 Marks)
Q3: The work function of caesium metal is 2.14 eV. If light of frequency $6 \times 10^{14}\text{ Hz}$ is incident on the metal, calculate the maximum kinetic energy of the emitted electrons.
Ans:
Given: $\phi_0 = 2.14 \text{ eV}$, $\nu = 6 \times 10^{14}\text{ Hz}$.
First, calculate the energy of the incident photon: $E = h\nu$.
$E = (6.63 \times 10^{-34} \text{ J s}) \times (6 \times 10^{14}\text{ Hz}) = 3.98 \times 10^{-19}\text{ J}$.
Convert this to eV: $\frac{3.98 \times 10^{-19}}{1.6 \times 10^{-19}} \approx 2.49 \text{ eV}$.
Using Einstein’s equation: $K_{max} = E – \phi_0$.
$K_{max} = 2.49 \text{ eV} – 2.14 \text{ eV} = 0.35 \text{ eV}$.
Q4: Show that the wavelength of electromagnetic radiation is equal to the de Broglie wavelength of its quantum (photon).
Ans:
For a photon, the momentum is given by $p = \frac{h\nu}{c}$.
Rearranging this, we get $\frac{h}{p} = \frac{c}{\nu}$.
We know from wave equations that $\frac{c}{\nu} = \lambda$ (wavelength of EM radiation).
According to de Broglie, the matter wave wavelength is $\lambda = \frac{h}{p}$.
Hence, both wavelengths are exactly equal.
Long Answer Type (5 Marks)
Q5: Describe the experimental study of the photoelectric effect. Explain the effect of (i) intensity of light on photoelectric current and (ii) potential difference on photoelectric current, using suitable graphs.
Ans:
Experimental Setup: The apparatus consists of an evacuated quartz/glass tube with a photosensitive emitter plate (C) and a collector plate (A). Monochromatic light passes through a quartz window and hits plate C, emitting electrons. A battery supplies a variable potential difference, and a microammeter measures the resulting photocurrent.
(i) Effect of Intensity: By keeping frequency and positive potential fixed, increasing the intensity of the light results in a linear increase in the photoelectric current. This is because higher intensity means more photons striking the surface per second, which ejects more photoelectrons per second. (Draw Figure-1 from the notes here).
(ii) Effect of Potential Difference: Keeping intensity and frequency fixed, applying a positive potential to plate A attracts electrons, increasing current until it maxes out at the “Saturation Current”. Reversing the polarity (making plate A negative) repels electrons. The current drops rapidly until it reaches zero at a specific negative voltage called the “Stopping Potential” ($V_0$). The stopping potential is independent of the light’s intensity. (Draw Figure-2 from the notes here).
Case-Based / Competency-Based Question (4 Marks)
Q6: In a research lab, a scientist named Rahul is testing a new photosensitive material for a solar cell. He illuminates the material with an Argon laser producing light of wavelength 488 nm. The stopping potential recorded is 0.38 V.. Later, he tries illuminating the same material with a red laser (wavelength 632.8 nm) but observes no photocurrent at all.
(a) Calculate the work function of the material from Rahul’s first experiment. (Take $h = 6.63 \times 10^{-34}\text{ J s}$, $c = 3 \times 10^8\text{ m/s}$, $e = 1.6 \times 10^{-19}\text{ C}$).
(b) Why did the red laser fail to produce any photocurrent?
Ans:
(a) We use Einstein’s equation: $eV_0 = \frac{hc}{\lambda} – \phi_0$.
Rearranging for work function: $\phi_0 = \frac{hc}{\lambda} – eV_0$.
Energy of incident light $\frac{hc}{\lambda} = \frac{(6.63 \times 10^{-34}) \times (3 \times 10^8)}{488 \times 10^{-9}}\text{ J} = 4.07 \times 10^{-19}\text{ J}$.
Convert to eV: $\frac{4.07 \times 10^{-19}}{1.6 \times 10^{-19}} = 2.54 \text{ eV}$.
Stopping potential energy $eV_0 = 0.38 \text{ eV}$.
Therefore, $\phi_0 = 2.54 \text{ eV} – 0.38 \text{ eV} = 2.16 \text{ eV}$.
(b) Red light has a longer wavelength than the argon laser (488 nm). Since Energy $E = \frac{hc}{\lambda}$, a longer wavelength means a lower photon energy. The energy of the red laser photons is less than the work function (2.16 eV) of the material. Therefore, its frequency is below the threshold frequency, resulting in no photoelectric emission.
Assertion-Reasoning Type (1 Mark)
Q7:
Assertion (A): The maximum kinetic energy of emitted photoelectrons depends on the frequency of the incident light, but not on its intensity.
Reason (R): The photoelectric effect happens because of a continuous absorption of energy spread over the entire wavefront of the incident light.
Ans: Option (c). Assertion is True, but Reason is False.
Explanation: The assertion is an experimentally proven fact derived from the stopping potential vs. frequency graph. However, the reason describes the classical wave theory, which entirely fails to explain the photoelectric effect. The correct reason is that emission is due to discrete, one-to-one photon-electron collisions.
End of Notes.
Students, practice the mathematical calculations involving Planck’s constant ($h$) carefully, as the powers of 10 can get tricky in exams. Also, memorize the definitions of Work Function, Threshold Frequency, and Stopping Potential flawlessly.
Read Also:
Chapter 10- Wave Optics
For more check official website of
NCERT
