1. Introduction: Looking at the World Around Us
Hello students! Look around your room right now. What do you see? Perhaps a wooden chair, a plastic pen, a glass of water, or the air you are breathing. In previous classes, we learned that anything that occupies space and has mass is called matter.
But have you ever wondered what this matter is built from? Is the water in your bottle just one single thing, or is it a combination of tinier particles? Is the air just empty space, or is it a soup of different gases?
In this chapter, we are going to put on our “detective glasses” and categorize every material in the universe into three main buckets:
- Elements (The simplest building blocks)
- Compounds (Chemical combinations)
- Mixtures (Physical combinations)
Understanding this is like learning the alphabet of the universe. Once you know the letters (elements), you can make words (compounds) and sentences (mixtures)!
2. What Exactly is a Mixture?
Let’s start with something delicious. Imagine a bowl of sprout salad or bhel puri. You have puffed rice, peanuts, onions, tomatoes, and spices. Even if you mix them thoroughly, you can still pick out a peanut or a piece of tomato, right?
This is exactly what a mixture is in science. A mixture is formed when two or more substances are mixed together physically, but they do not react chemically. This means each ingredient keeps its own original properties. The peanut is still a peanut; the puffed rice is still puffed rice.
Common examples of mixtures in our daily life include:
- Air: A mix of nitrogen, oxygen, carbon dioxide, dust, and water vapour.
- Lemonade: Water, sugar, lemon juice, and salt mixed together.
- Soil: A mix of sand, clay, decaying leaves, and pebbles.

Figure-1: A sprout salad is a classic example of a mixture. You can physically see and separate the different ingredients.
3. Types of Mixtures: Uniform vs. Non-Uniform
Not all mixtures look the same. Scientists classify them into two types based on how well the components are mixed.
A. Non-Uniform Mixtures (Heterogeneous)
In these mixtures, you can easily see the different particles. The composition is not the same throughout. If you take a spoonful of soil from one spot and a spoonful from another, they might have different amounts of sand or pebbles.
Examples: Oil and water, sand and salt, or the sprout salad we discussed earlier.
B. Uniform Mixtures (Homogeneous)
These are tricky! They are mixed so perfectly that the particles are evenly distributed. You cannot see the separate ingredients even with a microscope. The mixture looks like one single substance.
Examples: Sugar dissolved in water, clean air, or alloys like brass.
Teacher’s Note: Is Stainless Steel a pure metal? No! It is actually a uniform mixture (an alloy) of Iron, Nickel, Chromium, and Carbon. We mix them to prevent rusting and increase strength. Ancient India also had a rich tradition of making alloys like Bronze (Kamsya) and Brass.
4. The “Pure” vs. “Scientific Pure” Confusion
When you go to the grocery store, you see packets labeled “Pure Ghee,” “Pure Milk,” or “Pure Spices.” In everyday language, “pure” means it is not adulterated or spoiled.
However, to a scientist, none of these are pure!
- Milk is a mixture of water, fats, and proteins.
- Air is a mixture of gases.
Scientific Definition of Pure Substance: In science, a substance is “pure” only if it is made up of only one type of particle (atoms or molecules). It cannot be separated into other kinds of matter by physical methods like filtering or boiling.
5. Elements: The Building Blocks of the Universe
If you keep breaking down matter into smaller and smaller pieces, you eventually reach a point where you cannot break it down any further chemically. These basic substances are called Elements.
Think of elements as the “Lego bricks” of the universe. There are only about 118 known types of Lego bricks (elements), but they combine to build everything—from stars to smartphones.
Elements consist of only one type of atom. We classify them generally into:
- Metals: Iron, Gold, Silver, Copper (usually shiny, hard, conduct electricity).
- Non-Metals: Oxygen, Hydrogen, Carbon, Sulfur (can be solids or gases, usually brittle or invisible).
- Metalloids: Silicon, Boron (have mixed properties).
6. Compounds: When Elements Combine Chemically
When two or more elements combine chemically in a fixed ratio, they form a Compound. This is where the magic of chemistry happens!
Key Characteristic: The properties of a compound are completely different from the elements that made it.
The Water Paradox:
Consider Water ($H_2O$). It is made of:
1. Hydrogen: A gas that burns explosively (fuel).
2. Oxygen: A gas that supports burning.
But when they combine, they form Water, which is a liquid used to put out fire! This drastic change in property proves water is a compound, not just a mixture.

Figure-2: In a compound like water, atoms of Hydrogen and Oxygen are tightly bonded in a fixed ratio of 2:1. You cannot separate them by simple filtering.
7. Investigating Water: The Electrolysis Experiment
How do we prove water is made of Hydrogen and Oxygen? We can break it using electricity. This process is called Electrolysis.
The Experiment Setup:
- Take water in a beaker and add a few drops of acid (to help electricity flow).
- Place two test tubes filled with water over the terminals of a 9V battery inside the water.
- Watch carefully! You will see bubbles rising in the test tubes.
The Observation:
- Gases collect in the top of the test tubes, displacing the water.
- One test tube has double the volume of gas compared to the other.
The Confirmation Test:
- If you bring a burning candle near the tube with more gas, it makes a “POP” sound. This confirms it is Hydrogen.
- If you bring a glowing splinter near the other tube, it burns brighter. This confirms it is Oxygen.
This experiment confirms water is a compound of Hydrogen and Oxygen in a 2:1 ratio.
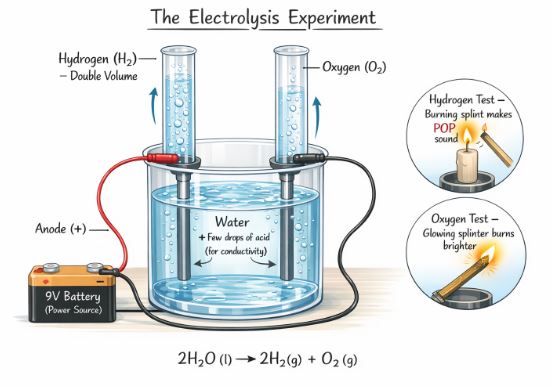
Figure-3: Passing electricity breaks the chemical bonds in water, separating it back into Hydrogen and Oxygen gases.
8. Investigating Sugar: Decomposition
What happens if you heat sugar in a test tube? It doesn’t just melt; it undergoes a chemical change.
- First, it turns brown, then black.
- Steam (water vapour) comes out and condenses on the cool upper parts of the tube.
- A black substance called Charcoal (Carbon) is left behind.
This proves that sugar is a compound made of Carbon, Hydrogen, and Oxygen. The heat broke the compound apart.
9. The Great Debate: Mixture vs. Compound (Iron and Sulfur)
This is one of the most important experiments in Class 8 Chemistry to understand the difference between mixing and reacting.
Case A: Just Mixing (Physical Change)
We take Iron filings (grey powder) and Sulfur powder (yellow) and just mix them in a bowl.
- Appearance: You can see yellow and grey specks.
- Magnet Test: If you bring a magnet near, the iron particles jump out and stick to the magnet. The components can be separated.
- Conclusion: This is a Mixture. No new substance is formed.
Case B: Heating the Mix (Chemical Change)
Now, we heat this mixture strongly until it glows red.
- Appearance: A black, hard mass is formed. You can no longer see separate yellow or grey particles.
- Magnet Test: The magnet has no effect. The iron has lost its magnetic property because it is chemically bonded.
- Chemical Test: If we add acid, it produces a gas with a “rotten egg” smell ($H_2S$), not the hydrogen gas produced by pure iron.
- Conclusion: This is a Compound called Iron Sulfide ($FeS$). A chemical reaction has occurred.

Figure-4: (Left) A magnet separates iron from the mixture. (Right) After heating, a new compound Iron Sulfide is formed which is not magnetic.
10. Minerals and Natural Resources
The earth beneath our feet is a treasure chest of chemical substances. Naturally occurring substances found in the earth’s crust are called Minerals.
- Most minerals are compounds (e.g., Quartz, Calcite, Iron Oxide).
- A few are native elements (e.g., Gold, Silver, Diamond/Carbon).
We use these minerals to extract metals, make cement, create jewelry, and even manufacture the talcum powder you use in summer (from the mineral Talc!).
11. Summary: Key Differences Table
Let’s summarize the main differences between Mixtures and Compounds to ensure we are exam-ready.
| Property | Mixture | Compound |
|---|---|---|
| Formation | Physical mixing. No new substance formed. | Chemical reaction. entirely new substance formed. |
| Composition | Variable (can change ratio of ingredients). | Fixed (elements combined in fixed ratio). |
| Properties | Shows properties of its components. | Properties are different from components. |
| Separation | Possible by physical methods (filtration, magnet). | Only possible by chemical methods (electrolysis). |
Practice Questions (CBSE Pattern)
A. Very Short Answer Questions
Q1: Define the term ‘Element’.
Answer: An element is a pure substance made up of only one type of atom that cannot be broken down into simpler substances by chemical means.
Q2: Is the “pop” sound observed during the testing of Oxygen or Hydrogen?
Answer: Hydrogen gas burns with a “pop” sound.
Q3: Name the method used to separate Hydrogen and Oxygen from water.
Answer: Electrolysis.
B. Short Answer Questions
Q1: Why is air considered a mixture and not a compound?
Answer: Air is a mixture because its components (nitrogen, oxygen, etc.) retain their individual properties, they are not chemically combined, and their proportion varies from place to place.
Q2: Give two examples of Uniform Mixtures and two examples of Non-Uniform Mixtures.
Answer: Uniform: Salt solution, Stainless steel. Non-Uniform: Sand and water, Sprout salad.
Q3: What happens when sugar is heated strongly in a test tube?
Answer: Sugar decomposes (breaks down) into a black substance called carbon (charcoal) and releases water vapour. This proves sugar is a compound.
C. Long Answer Questions
Q1: Describe an activity to show the difference between a mixture of Iron and Sulfur and the compound Iron Sulfide.
Answer:
1. Mixture: Mix Iron filings and Sulfur powder. Bring a magnet near it. The iron is attracted and separated. This shows components retain properties in a mixture.
2. Compound: Heat the mixture until it becomes a black mass ($FeS$). Bring a magnet near it. Iron is not attracted. This shows the compound has new properties and cannot be separated physically.
Q2: Explain the electrolysis of water with a labeled diagram description.
Answer: When electric current is passed through acidified water using a battery and electrodes, water breaks down into Hydrogen and Oxygen gases. Hydrogen collects at the negative terminal and Oxygen at the positive terminal. The volume of Hydrogen collected is double that of Oxygen (Ratio 2:1).
D. Case-Based / Competency Question
Case: Rohan found a shiny yellow rock in his garden. He thought it was gold. His teacher told him to heat it. Upon heating, the rock released a gas that smelled like burning sulfur, and a black residue was left.
Q1: Was the rock an element or a compound? Why?
Answer: It was likely a compound (a mineral containing sulfur) because it broke down into simpler substances (gas and black residue) upon heating. Elements do not decompose on simple heating.
E. Assertion–Reason
Assertion: Water is a compound, but tap water is a mixture.
Reason: Tap water contains dissolved salts and minerals in addition to pure water molecules.
Answer: Both Assertion and Reason are true, and Reason is the correct explanation for Assertion. Pure $H_2O$ is a compound, but tap water has impurities mixed in it physically.
Read Also
For official syllabus and textbooks, visit the
NCERT Official Website.

