
Start Chapter MCQ Quiz
📘 Download Chapter Notes (PDF)
Detailed Guide: Electric Charges and Fields Class 12 Physics
1. Introduction: The Spark of Curiosity
Hello everyone! Welcome to Class 12 Physics.
Electric Charges and Fields Class 12 Physics is Chapter 1 of electrostatics and forms the foundation of electricity and magnetism. We are starting a fascinating journey today into Electromagnetism, a force that literally holds the world together. We will begin with the simplest case: charges that are sitting still. This branch of physics is called Electrostatics.
You have experienced electrostatics countless times in your daily life. Have you ever pulled a woollen sweater over your head in winter and heard a crackling sound? Or perhaps you’ve seen a spark in the dark while doing so? Have you ever rubbed a balloon on your hair and stuck it to a wall? Or shuffled your feet on a carpet and felt a “zap” when touching a metal doorknob? These aren’t just magic tricks; they are evidence of electric charge.
“Static” means stationary. So, in this chapter, we are studying the forces, fields, and potentials associated with charges that are at rest. This is the foundation for everything else—from the current running in your home wiring to the complex signals inside a computer processor.
2. Electric Charge: The Basics
Historically, the study of electricity began with a simple observation. Around 600 BC, Thales of Miletus found that amber (a fossilized tree resin) rubbed with wool attracted light objects like dry leaves. The Greek name for amber is elektron, which gives us the modern word “Electricity”.
2.1 Two Kinds of Charges
Through rigorous experimentation in the 18th century, scientists discovered a crucial duality in nature. They found that if you rub a glass rod with silk and a plastic rod with fur, they behave differently.
- If you bring two glass rods (rubbed with silk) near each other, they repel.
- If you bring two plastic rods (rubbed with fur) near each other, they also repel.
- BUT, if you bring a charged glass rod near a charged plastic rod, they attract each other.
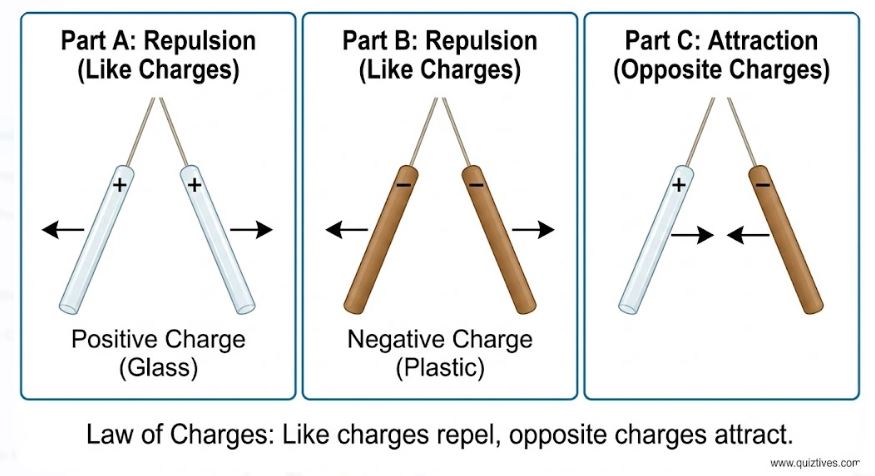
Benjamin Franklin proposed a convention that we still use today. He called the charge on the glass rod Positive and the charge on the plastic rod Negative. If an object has no net charge, we say it is Electrically Neutral. It is important to remember that “positive” and “negative” are just names; there is nothing inherently “minus” about negative charge, other than its mathematical property of canceling out positive charge.
2.2 Conductors and Insulators
Why does a metal spoon not get charged when you rub it? It actually does! But because metal is a Conductor, the charge flows through the spoon, through your body, and into the earth instantly. Conductors (like metals, human body, earth) allow charge to flow freely because they possess “free electrons” that are not bound to any specific atom.
Insulators (like glass, plastic, wood) do not allow charge to flow. The electrons are tightly bound to the nucleus. If you put a charge on a plastic comb, it stays right where you put it. This is why static electricity experiments work best with insulators.
3. Methods of Charging
How do we actually give an object a charge? There are three main ways to electrify a body.
3.1 Charging by Friction
This is the rubbing method we discussed. When you rub glass with silk, the friction provides enough energy to rip electrons off the glass atoms and transfer them to the silk. The object that loses electrons (glass) becomes Positive. The object that gains electrons (silk) becomes Negative. Note that mass is also transferred (since electrons have mass), but the amount is negligible.
3.2 Charging by Conduction
This involves physical contact. If you touch a charged metal sphere to an uncharged metal sphere, the repulsive forces between the like charges push some of them onto the new sphere. The charge is shared between the two bodies.
3.3 Charging by Induction
This is the coolest method because there is no physical contact involved. It uses the influence of a nearby charge to redistribute electrons.
1. Bring a negatively charged rod near a neutral metal sphere.
2. The free electrons in the sphere are repelled to the far side, leaving the near side positive. This is called polarization.
3. Connect the far side to the ground (Earth). The repulsive force drives the electrons down the ground wire.
4. Remove the ground connection, then remove the rod. The sphere is left with a net positive charge!
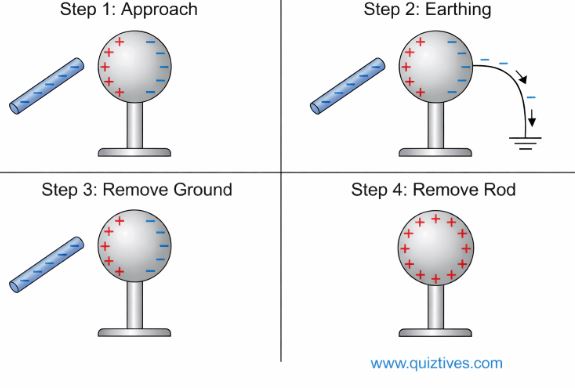
4. Basic Properties of Electric Charge
Whatever the type of charge, it follows three fundamental rules. These are the pillars of electrostatics.
4.1 Additivity of Charge
Electric charge is a scalar quantity. It has magnitude but no direction. It adds up like normal numbers. If a system contains multiple charges q1, q2, q3, the total charge Q is simply the algebraic sum. You must include the signs.
Example: If a system has charges +2 C and -5 C, the total charge is -3 C.
4.2 Conservation of Charge
Charge is neither created nor destroyed. It is only transferred. When you rub a balloon on your hair, you aren’t creating charge; you are moving electrons from your hair to the balloon. The total charge of the (Hair + Balloon) isolated system remains zero. This law holds true even in nuclear reactions and high-energy physics.
4.3 Quantization of Charge
This is a non-intuitive property. We tend to think of charge as a fluid, like water. But charge is actually grainy, like sand. The smallest possible charge that can exist independently is the charge on an electron or proton, denoted by ‘e’.
e = 1.602 × 10-19 C
Any charge ‘q’ on a body is always an integral multiple of ‘e’.
q = n e
Where ‘n’ is an integer (1, 2, 3, -1, -2…). You cannot have a charge of 1.5e or 2.7e. This was experimentally proved by Millikan in his famous Oil Drop Experiment.
Why don’t we feel the “graininess”?
At the macroscopic level (daily life), the charge ‘e’ is so tiny that the quantization is invisible. A charge of just 1 micro-Coulomb (μC) contains about 6 trillion electrons! Adding or removing one electron doesn’t make a noticeable difference. We treat charge as continuous in macroscopic calculations, just like we treat water as a fluid even though it’s made of discrete molecules.
5. Coulomb’s Law
We know like charges repel and unlike attract. But how strong is this force? Charles Augustin de Coulomb measured this in 1785 using a torsion balance. He found that the electric force behaves very much like gravity, but it is vastly stronger.
Statement: The force between two point charges is directly proportional to the product of their magnitudes and inversely proportional to the square of the distance between them. The force acts along the line joining the centers of the two charges.
F = k (q1 q2) / r2
Here, ‘k’ is the electrostatic constant.
In vacuum (or air), k = 9 × 109 N m2/C2.
In physics, we usually write ‘k’ in a more fundamental form:
k = 1 / (4 π ε0)
Here, ε0 (Epsilon Naught) is called the Permittivity of Free Space. It represents how easily the vacuum permits electric field lines to pass through.
Value of ε0 = 8.854 × 10-12 C2/N m2.
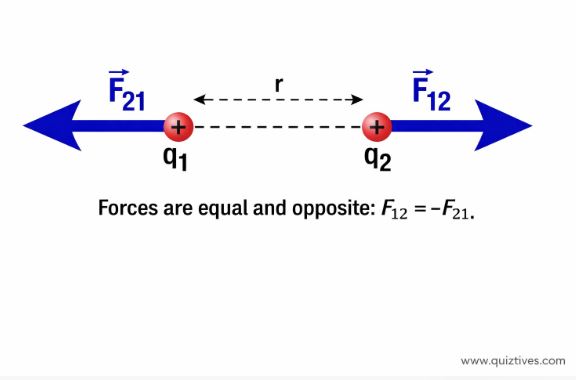
Vector Form
Since Force is a vector, we need to specify direction.
F12 = Force on charge 1 due to charge 2.
F12 = [ 1 / (4 π ε0) ] × [ (q1 q2) / r2 ] × unit vector r21
This vector notation automatically handles the direction: if q1q2 > 0 (repulsion), the force is outward. If q1q2 < 0 (attraction), the force is inward.
New Force F’ = F / K
Here, ‘K’ is the Dielectric Constant (or Relative Permittivity εr) of the medium. For water, K is about 80. This means the electric force between ions in water is 80 times weaker than in air! This is why salt (NaCl) dissolves so easily in water—the water weakens the bond between Na+ and Cl-.
6. Electric Field
Here is a deep question: How does charge A know charge B is there? They aren’t touching. This “Action at a Distance” puzzled scientists for a long time. Michael Faraday solved this by introducing the concept of a Field.
Imagine that every charge creates an “aura” or “field” in the space around it. When another charge enters this field, the field exerts a force on it. The field is the mediator. Charge A creates the field, and the field pushes Charge B.
Electric Field (E) = Force (F) / Test Charge (q0)
E is a vector quantity. Its direction is the same as the direction of the force experienced by a positive test charge.
SI Unit: Newton/Coulomb (N/C).
Field due to a Point Charge
Using Coulomb’s law, we can derive the field ‘E’ at a distance ‘r’ from a source charge ‘Q’:
E = (1 / 4 π ε0) × (Q / r2)
Notice that ‘E’ depends only on the source charge Q and distance r. It does not depend on the test charge. If Q is positive, E points radially outward. If Q is negative, E points radially inward.
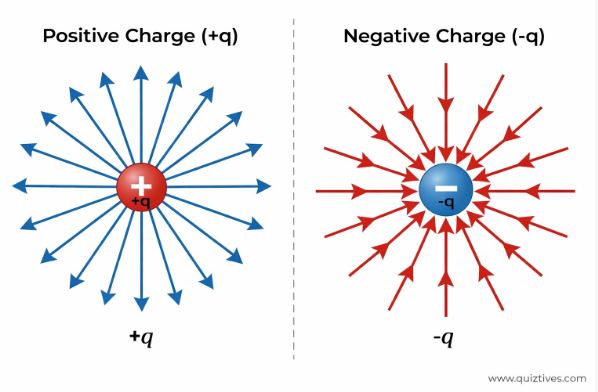
Electric Field Lines
Field lines are an imaginary geometrical way to visualize the electric field. They are not real wires in space, but they help us map the force.
Key Properties of Field Lines:
1. They always start from Positive charges and end at Negative charges.
2. They never cross each other. (If they did, the field would have two directions at one point, which is physically impossible).
3. The tangent to the line at any point gives the direction of E.
4. Crowded lines indicate a Strong Field; spaced out lines indicate a Weak Field.
5. They do NOT form closed loops (unlike magnetic field lines). This is because the electrostatic field is “conservative”.
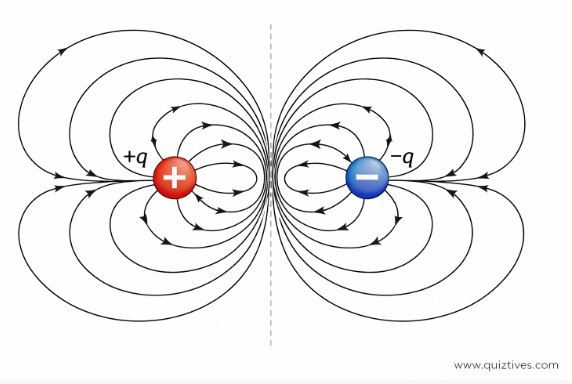
7. Electric Dipole
An Electric Dipole is a pair of equal and opposite charges (+q and -q) separated by a small distance (2a). This might seem like a specific textbook case, but it’s very real. Many molecules, like water (H2O) and hydrochloric acid (HCl), are natural dipoles.
We define a vector called the Dipole Moment (p) to measure the strength of the dipole.
Direction: By convention, from Negative (-q) to Positive (+q).
p = q × 2a
Electric Field of a Dipole
The total field of a dipole is the vector sum of the fields from +q and -q. We derive the field at two specific positions:
1. On the Axial Line (End-on position):
For a point at distance r from the center (where r >> a):
Eaxial = (2k p) / r3
2. On the Equatorial Line (Broad-side on position):
For a point at distance r from the center (where r >> a):
Eequatorial = (k p) / r3
Important Observation: The field of a point charge drops off as 1/r2, but the field of a dipole drops off faster, as 1/r3.
Dipole in a Uniform External Field
What happens if you place a dipole in a uniform electric field E?
The +q charge feels a force qE in the direction of the field.
The -q charge feels a force qE opposite to the field.
The Net Force is Zero. The dipole won’t move forward or backward.
However, these two forces act at different points, creating a turning effect or Torque (τ). This torque tries to rotate the dipole to align it with the field.
τ = p E sin(θ)
In vector form: τ = p × E (Cross Product).
8. Electric Flux and Gauss’s Law
This section introduces one of the most powerful tools in electrostatics, developed by the mathematical genius Carl Friedrich Gauss.
Electric Flux (Φ)
Imagine water flowing through a net. “Flux” is a measure of how much water passes through. Similarly, Electric Flux is a measure of the “flow” of electric field lines passing through a given area.
Φ = E × A × cos(θ)
Where:
E = Electric Field intensity
A = Area of the surface
θ = Angle between the Electric Field vector and the Area Vector.
Note: The Area Vector is always perpendicular (normal) to the surface.
SI Unit: N m2/C.
Gauss’s Law
Gauss found a beautiful relationship between the total flux flowing out of a closed surface and the charge enclosed inside it.
Statement: The total electric flux through a closed surface is equal to (1 / ε0) times the net charge enclosed by the surface.
Φ = ∮ E . dA = qenclosed / ε0
This law is incredibly useful. It allows us to calculate the Electric Field of symmetric objects (like spheres, cylinders, and sheets) in just a few lines of math, where Coulomb’s law would require pages of complex integration.
Applications of Gauss’s Law
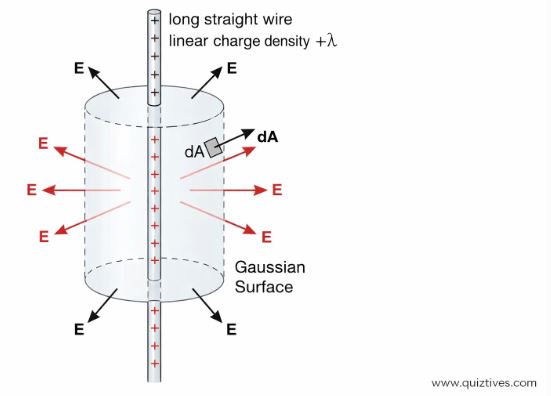
1. Field due to an Infinitely Long Straight Wire
Imagine a long wire with linear charge density λ (C/m). We imagine a cylindrical Gaussian surface around it. By symmetry, the field is radial.
E = λ / (2 π ε0 r)
Here, E is inversely proportional to r (distance from wire).
2. Field due to an Infinite Plane Sheet
For a flat sheet with surface charge density σ (C/m2), the field is uniform and directed away from the sheet.
E = σ / (2 ε0)
Notice something strange? The field does not depend on distance r. It is constant everywhere near the sheet!
3. Field due to a Thin Spherical Shell
Consider a hollow shell with charge Q.
Outside the shell (r > R): It behaves as if all charge is concentrated at the center. E = kQ / r2.
Inside the shell (r < R): The Electric Field is ZERO.
This phenomenon is used in Electrostatic Shielding. If you are inside a hollow conductor (like a car or a metal cage) during a lightning storm, you are safe because the electric field inside remains zero.
9. Practice Questions with Solutions
Physics is best learned by doing. Let’s apply these concepts to some problems.
Q1: Quantization of Charge
Question: A polythene piece rubbed with wool is found to have a negative charge of 3 × 10-7 C. Estimate the number of electrons transferred.
Solution:
We know q = n e.
Here, q = 3 × 10-7 C and e = 1.6 × 10-19 C.
n = q / e
n = (3 × 10-7) / (1.6 × 10-19)
n = 1.875 × 1012.
Since n must be an integer, we round it to approximately 2 trillion electrons.
These electrons transferred from wool to polythene (since polythene became negative).
Q2: Coulomb’s Law
Question: Two small spheres spaced 20 cm apart have charges +2 μC and +6 μC. Find the force between them. If they are touched and placed back, what is the new force?
Solution:
Initial Force:
F = k (q1 q2) / r2
F = (9 × 109) × (2 × 10-6) × (6 × 10-6) / (0.2)2
F = (108 × 10-3) / 0.04
F = 2.7 N (Repulsive).
After Touching:
Charge is shared equally. Total Q = 2 + 6 = 8 μC.
Each sphere gets 4 μC.
F’ = (9 × 109) × (4 × 10-6) × (4 × 10-6) / (0.2)2
F’ = (144 × 10-3) / 0.04
F’ = 3.6 N.
Result: The repulsive force increased!
Q3: Flux Calculation
Question: A point charge of 10 μC is at the center of a cubic Gaussian surface of edge 10 cm. What is the net flux through the surface?
Solution:
According to Gauss’s Law, the total flux depends only on the enclosed charge, not the shape or size of the box.
Φ = q / ε0
Φ = (10 × 10-6) / (8.854 × 10-12)
Φ ≈ 1.13 × 106 N m2/C.
Read Also
For official syllabus and textbooks, visit the
NCERT Official Website.
